General Information On CTLA-4 protein
CTLA-4 protein (cytotoxic T-lymphocyte-associated protein 4) also known as Cluster of Differentiation 152 (CD152) is a protein receptor that acts as an immune checkpoint. CD152 protein is expressed in regulatory T cells. CTLA-4 protein is upregulated following T cell activation.
CTLA-4 protein is homologues to CD28 protein although they have opposite functions. CTLA-4 protein transmits inhibitory signals to T cells hence acting as ‘off’ switch whereas CD28 is a T-cell co-stimulatory protein. They both bind to CD80 and CD86 on antigen presenting cells. Furthermore, CD152 bind to CD80 and CD86 with a greater affinity than CD28 and surpasses CD28 activity on the ligands. Research data (Stamper et al. 2001) showed that CTLA-4 and CD80 a periodic arrangement in which bivalent CTLA-4 homodimers bridge bivalent CD80 homodimers. It is believed that this oligomerization provides the structural basis for forming unusually stable signaling complexes at the T-cell surface.
The structure of CTLA-4 protein consist in an extracellular V domain, a transmembrane domain and a cytoplasmic tail. The membrane-bound isoform functions as a homodimer interconnected by a sulfide bond whereas the soluble isoform functions as a monomer. CTLA-4 protein has an intracellular domain which is believed to have no intrinsic catalytic activity. This intracellular domain, similar to that of CD28, contains one YVKM motif that binds PI3K, PP2A, SHP-2 and one proline rich motif that binds to SH3 containing proteins. It is believed that CTLA-4 inhibits T cell activity through SHP-2 and PP2A dephosphorylation of TCR-proximal signaling proteins (CD3 and LAT).
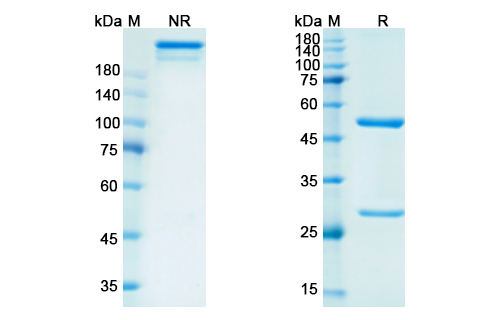
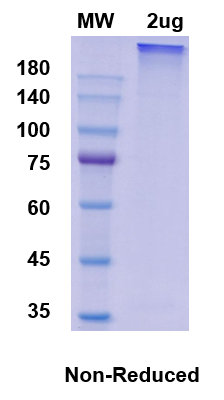
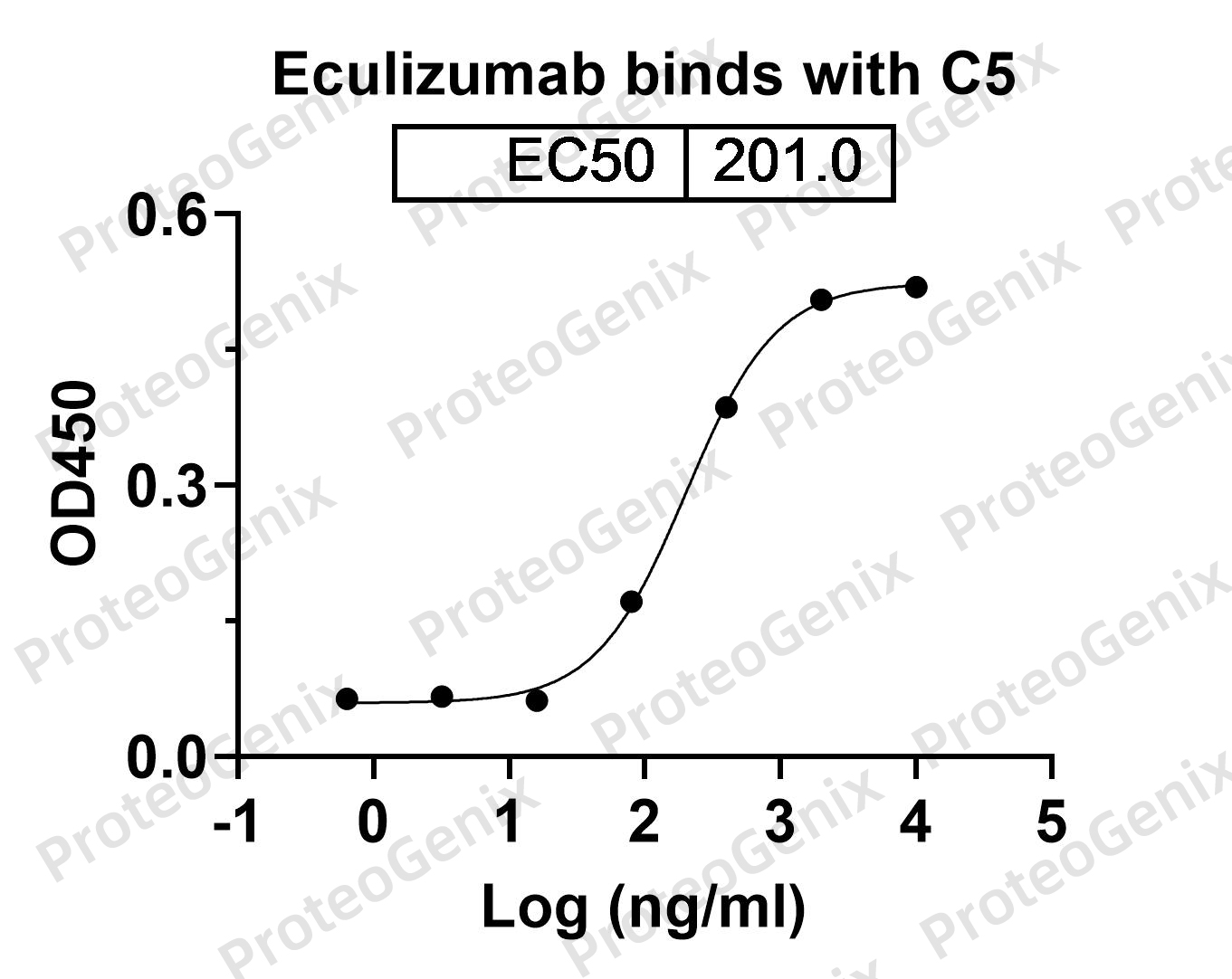
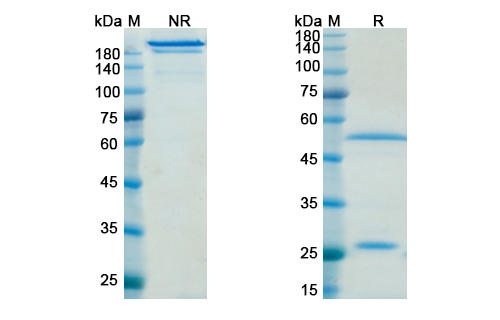
There are no reviews yet.