Introduction
Labetuzumab Biosimilar, also known as Anti-CEACAM5 mAb, is a monoclonal antibody that has been developed as a biosimilar to the original Labetuzumab. This antibody specifically targets the carcinoembryonic antigen-related cell adhesion molecule 5 (CEACAM5), which is overexpressed in various types of cancer. In this article, we will discuss the structure, activity, and potential applications of Labetuzumab Biosimilar in the field of cancer research.
Structure of Labetuzumab Biosimilar
Labetuzumab Biosimilar is a recombinant, humanized IgG1 monoclonal antibody that has been engineered to have a similar structure to the original Labetuzumab. It consists of two heavy chains and two light chains, connected by disulfide bonds. The heavy chains contain constant regions (Fc) and variable regions (Fab), while the light chains contain only variable regions. The variable regions are responsible for binding to the target antigen, CEACAM5, while the constant regions are involved in effector functions such as antibody-dependent cellular cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC).
Activity of Labetuzumab Biosimilar
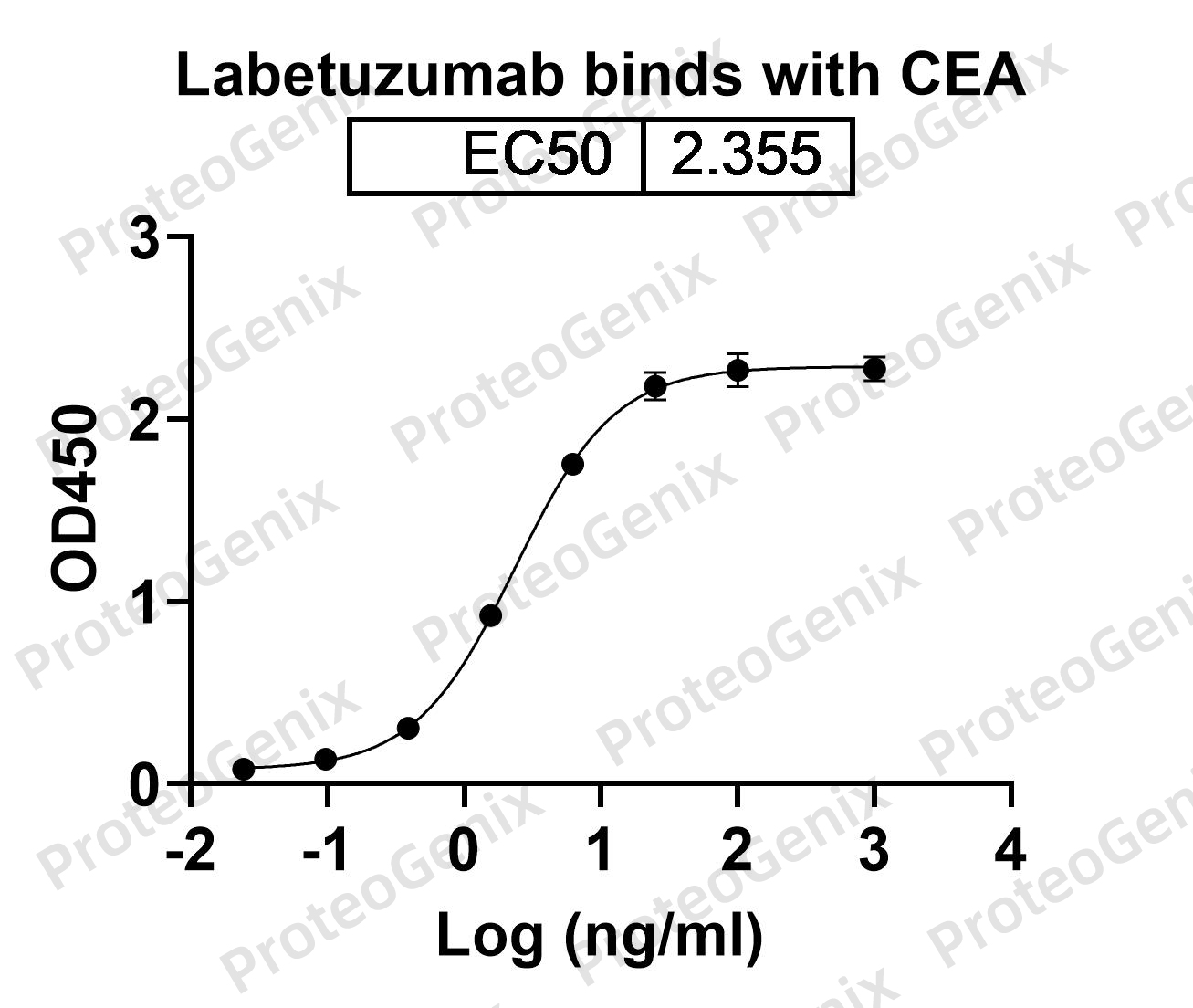
Labetuzumab Biosimilar binds specifically to CEACAM5, a glycoprotein that is overexpressed in a variety of cancers, including colorectal, pancreatic, and lung cancer. This binding inhibits the growth and spread of cancer cells by blocking the interaction between CEACAM5 and its ligands, which are involved in cell adhesion and signaling pathways. Additionally, Labetuzumab Biosimilar can induce ADCC and CDC, leading to the destruction of cancer cells by the immune system.
Potential Applications of Labetuzumab Biosimilar
Labetuzumab Biosimilar has shown promising results in preclinical and clinical studies as a potential therapeutic agent for various types of cancer. Here are some potential applications of this antibody:
1. Treatment of Colorectal
Cancer CEACAM5 is highly expressed in colorectal
cancer cells, making it an ideal target for Labetuzumab Biosimilar. In a phase II clinical trial, Labetuzumab Biosimilar showed promising results in combination with chemotherapy in patients with metastatic colorectal cancer. It significantly improved progression-free survival and overall survival compared to chemotherapy alone.
2. Treatment of Pancreatic
Cancer Pancreatic
cancer is one of the most aggressive and difficult-to-treat cancers. CEACAM5 is overexpressed in pancreatic cancer cells, and Labetuzumab Biosimilar has shown efficacy in preclinical studies. A phase I clinical trial is currently ongoing to evaluate the safety and efficacy of Labetuzumab Biosimilar in combination with chemotherapy in patients with advanced pancreatic cancer.
3. Treatment of Lung
Cancer CEACAM5 is also overexpressed in lung
cancer cells, and Labetuzumab Biosimilar has shown promising results in preclinical studies. A phase II clinical trial is currently ongoing to evaluate the safety and efficacy of Labetuzumab Biosimilar in combination with chemotherapy in patients with advanced non-small cell lung cancer.
4. Research Tool for
Cancer Studies
In addition to its potential therapeutic applications, Labetuzumab Biosimilar can also serve as a valuable research tool for studying the role of CEACAM5 in cancer development and progression. It can be used in various techniques such as flow cytometry, immunohistochemistry, and Western blotting to detect and quantify CEACAM5 expression in cancer cells.
Conclusion
Labetuzumab Biosimilar is a promising biosimilar of the original Labetuzumab, with a similar structure and activity. Its specific targeting of CEACAM5, a protein overexpressed in various types of cancer, makes it a potential therapeutic agent for colorectal, pancreatic, and lung cancer. Furthermore, it can also serve as a valuable research tool for studying the role of CEACAM5 in cancer. Further clinical trials are needed to fully evaluate the efficacy and safety of Labetuzumab Biosimilar, but it holds great
There are no reviews yet.