General information on ACE2 protein
Angiotensin-converting protein also known as ACE2 is a zinc containing metalloprotein located on the surface of the cell of most organs including the lungs, arteries, heart, intestines, kidneys, cerebral cortex, brainstem, hypothalamus and striatum. It is a type I membrane protein whose enzymatic enzymatically active domain is located on the cell surface. The extracellular domain is cleaved by the action of another protein (known as sheddase) and is released in the blood stream. The main function of ACE2 protein is to decrease blood pressure by catalyzing hydrolysis of angiotensin II which is a constrictor peptide. Furthermore, ACE2 protein is regarded as an essential counter-regulatory carboxypeptidase of the renin-angiotensin hormone system. The latter is another critical regulator of blood volume and cardiovascular homeostasis.
ACE2 has also been identified as an entry point for some of the coronaviruses (CoV). CoV family of viruses are responsible for coronavirus disease (COVID) that can affect birds, animals including humans. More specifically COVID spike protein binds the enzymatic domain of ACE2 located on the cell surface which leads to virus entering the cell by endocytosis. Once in the cell, the virus is translocated int endosomes within the cell. Despite its implication in viral infection, ACE2 protein has shown protective effect against COVID by enhancing the production of the vasodilator angiotensin 1-7. This hypothesis is further supported the fact that ACE2-viral spiral protein interaction resulted in lower levels of ACE2 protein in mice. The reduced levels of ACE2 protein are believed to be due to its internalization alongside the viral particle.
ACE2 has also been involved in a number interaction that lead to the cleavage of other peptides such as apelin, dynorphin A, [des-Arg9]-bradykinin, , neurotensin, and ghrelin with high efficiency. Furthermore, ACE2 protein regulates the transportation of the neural amino acid transporter SLC6A19 across the membrane and has been implicated in Hartnup’s disease.
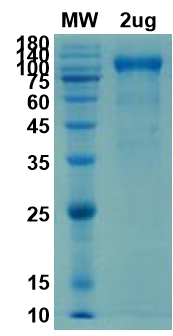
There are no reviews yet.