General information on TMPRSS2 protein
Transmembrane protease serine 2 (TMPRSS2) is a plasma membrane-anchored serine protease encoded by TMPRSS2 gene in humans. The protein consists of a type II transmembrane domain, a scavenger receptor cysteine-rich domain, a receptor class A domain, and a protease domain. The protein is regulated by androgen, a natural or synthetic hormone that plays a role in male traits and reproductivity. Androgens are often called “male hormones” although it is not exclusively found in males. TMPRSS2 regulates several substrates such as the protease activated receptor-2/F2RL1, pro-hepatocyte growth factor/HGF and matriptase/ST14. The activation of these substrates result in the disruption of extracellular matrix. TMPRSS2 protein is involved in both physiological and pathological processes. On one hand, the protein is involved in the proteolytic cascade that is responsible for the normal function of prostate. On the other hand, TMPRSS2 protein expression levels are increased by abnormal androgenic hormones activation in prostate cancer. That is not the case in androgen-independent prostate cancer tissue. TMPRSS2 also activates the trigeminal neurons which can contribute to spontaneous pain as well as mechanical allodynia.
It is believed that TMPRSS2 facilitates pathogenic Severe acute respiratory syndrome coronavirus 2 (SARS-coronavirus 2) entry in the cell. The latter is a coronavirus strain and a member the family Coronaviridae. The members of this family of viruses typically cause mild respiratory disease in humans. However, SARS-coronavirus 2 is responsible for novel coronavirus disease 2019 also known as COVID-19. SARS-coronavirus 2 entry in the target cell is facilitated by spike (S) protein. S protein facilitates viral cell entry by binding to cellular receptors and by priming the host cell proteases. S protein uses ACE2 receptor for entry and the TMPRSS2 serine protease for S protein priming. Without TMPRSS2 protein, target cells are significantly less sensitive to coronavirus but they are not entirely resistant to the virus as other host proteases such as cathepsins, can prime S protein.
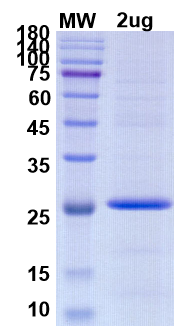
There are no reviews yet.