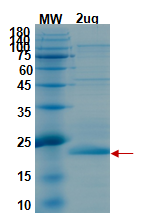
General information on NSP1
Non-structural protein 1 (NSP1) is a membrane-associated protein found in coronavirus. It is responsible for anchoring the viral replication complex to target cell membrane. Once the cell is infected, nsp1 protein interacts with 40S ribosomal subunit of the host which suppresses host gene expression. As a result, the viral particle is able to avoid host immune response which facilitates viral gene expression. Nsp1 protein has attracted attention following the recent emergence pathogenic human coronavirus (CoV). ). CoV can affect humans as well as animals. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) which is a CoV species is responsible for corona virus disease 2019 (COVID-19). CoV is an enveloped, single-stranded, positive-sense RNA genome whose length ranges from 27 to 32 kb. It is the largest genome known among RNA viruses. The viral genome has two large overlapping open reading frames (ORFs), ORF 1a and ORF 1b. Once in the cell, the viral genome produces two large precursor polyproteins 1a (pp1a) and 1ab (pp1ab) which are then processed into 16 nonstructural proteins (nsp1-nsp16). While nsp3–nsp16 share similar conserved functional domains, nsp1 sequence varies among different CoVs genera. Among the four known CoV genera, only α-CoVs and β-CoVs encode nsp1with no significant sequence similarity.
Nsp1 protein is regarded a potential virulence factor because of increasing evidence of its role in attenuating host innate immune response. As such nsp1 is considered a potential target to fight off coronavirus infection. There are functional conservation and mechanistic divergence observed among the nsp1 of different mammalian CoVs. In infected cells, nsp1 protein engages in a multi-pronged mechanism that eventually blocks the assembly of translationally competent ribosome which induces endonucleolytic cleavage and host mRNAs degradation. It is believed that nsp1 manipulates the nuclear pore complex which leads to nuclear-cytoplasmic shuttling disruption of the cellular proteins. There are multiple mutations on the surface residues of nsp1 that differentially inhibit gene expression.
There are no reviews yet.