Cart (0 Items)
Your cart is currently empty.
View ProductsIt looks like you are visiting from outside the EU. Switch to the US version to see local pricing in USD and local shipping.
Switch to US ($)
Whether you need ready-made naïve antibody libraries or a partner to develop high-diversity immune libraries for antibody phage display, ProteoGenix offers the market’s most extensive guarantees. Drawing from our experience in creating over 60 high diversity libraries (109 to 1010), our library generation process is designed to recover and optimize antibody repertoires from any host species (mouse, rabbit, camelid, human, etc.) and in any format (scFv, Fab, VHH) maximizing library diversity.
Use our naïve antibody libraries to quickly develop functional antibodies for therapy (3 fully human libraries) and analytical applications (rabbit library), as well as next-generation VHH antibodies in less than 7 weeks.
Develop functional and high-diversity immune antibody libraries (109-1010 different clones) even against the most challenging of antigens in only 14-19 weeks thanks to our vast expertise in antibody library generation processes.
Our optimized library generation protocols allow the construction of high-quality repertoires from any format, origin, or species for maximal clonal diversity (1010) and functionality adapted to any antigen and application
You get full ownership over your custom-made immune antibody libraries and binders obtained from our highly diverse naïve antibody libraries
Partner with us for your phage display projects or buy the libraries (immune or naïve) with all essential components to perform your own efficient panning campaigns
Drawing from our experience in library creation from any host species (mouse, sheep, llama, rabbit, etc.), we have developed optimal immunization and library generation protocols that prime your projects for success
Wide range of antibody formats to fit all types of applications
Minimize animal use in your antibody discovery projects by choosing our premium naïve libraries built for the highest possible quality and diversity
Our antibody phage display service process
Host selection & immunization*
*only for immune library generation
Cell harvesting
Antibody repertoire harvesting
Phagemid library construction
14-19 weeks
Library purchase
Buy our naïve or immune libraries to carry out phage display campaigns at your facilities
Phage display
Choose to perform your phage display panning campaigns with us
Antibody libraries can be generated from naïve hosts who have never been challenged with a specific immunogen or from immune hosts who have developed a strong and specific humoral response against the desired target. In comparison to other antibody generation approaches, antibody libraries have considerable advantages including:
Naïve antibody libraries typically comprise the vast naïve (unchallenged) antibody repertoire of many individual hosts to maximize diversity and functionality. In contrast, immune antibody libraries are built from a reduced number of hosts and maximize the affinity of the antibody repertoire against a specific immunogen (require animal immunization).
Antibodies generated from naïve libraries may, at times, present lower affinity towards a specific target in comparison to antibodies generated from immune libraries. But the choice between the two library types can be a complex one. It depends on several factors including the desired timeline, available budget, and project’s goals.
Naïve libraries are uniquely adapted to projects requiring fast turnarounds or projects focused on developing antibodies against different antigens (i.e. development of antibody cocktail therapies, antivenom treatments, etc.). Moreover, naïve library construction is generally considered more time-consuming and labor-intensive, these libraries can serve a wide diversity of projects in comparison to immune libraries, which are restricted to a single antigen. However, for projects that prize antibody affinity, the development of immune antibody libraries should be preferred.
| Naïve libraries | Immune libraries | |
|---|---|---|
| Species | Human, rabbit, camelids (camel, llama, alpaca) | No limitation except humans |
| Formats | Fab, scFv, VHH | Fab, scFv, VHH |
| Number of binders | ++ | +++ |
| Affinity of binders | ++ | +++ |
| Potential immunogenicity issues | No | yes |
| Animal use | No | yes |
| Timelines | 6-7 weeks | 14-19 weeks + 6-7 weeks |
| Price | + | ++ |
Thanks to its expertise, ProteoGenix is able to provide you with diverse high-quality naïve libraries generated through our unique platform of antibody library generation for phage display applications. Our libraries contain a vast repertoire of antibodies in many formats (scFv, Fab, or even VHH) and from many different species.
Overcome immunogenicity issues
Of a target
Short turnaround time
Example: 3-4 months from a naive library panning VS 9 months for standard hybridoma technology to get a purified recombinant antibody sample!
Animal protection
As phage display from naive libraries doesn’t require any animal use (unlike for immune library service or hybridoma development method)
Very high diversity of variants tested
We work on huge libraries of up to 1010 variants made of tens of donors and mixes of different strains/ethnic groups for maximized antibody repertoire.
No need for further humanization
Save lots of efforts, time and money thanks to our human naïve libraries, and greatly speed up your way to the clinic!
| Library | Format | Species | Size (clones) | Type of library |
|---|---|---|---|---|
| LiAb-SFCOVID-19TM | scFv | Human – Donors that recovered from COVID-19 – Ideal for COVID-19 antibody generation | 1.19 X 1010 | Immune |
| LiAb-SFCANCERTM Library | scFv & Fab | PBMCs & BMMCs from Human Donors (86 patients with 18 different cancers) including:
|
|
Immune |
| LiAb-SFAUTOIMMTM Library | scFv & Fab | PBMCs & BMMCs from Human Donors (57 patients with 12 different autoimmune diseases) including:
|
|
Immune |
| LiAb-SFMAXTM | scFv & Fab | Human – 5 different ethnic groups – 368 donors | 5.37 X 1010 | Naive |
| LiAb-SFaTM | scFv | Human | 1.5 X 109 | Naive |
| LiAb-FabTM | Fab | Human | 2.00 X 1010 | Naive |
| LiAb-SFDogTM | scFv & Fab | 46 healthy dogs from 6 different breeds: Beagle, German Shepherd, Labrador, English Coonhound, Great Dane, Chinese Rural Dog |
|
Naive |
| LiAb-SFCatTM | scFv & Fab | 52 healthy animals of 8 different breeds (Ragdoll, Maine Coon, Persian, Domestic Shorthair, Domestic Longhair, American Shorthair, Siamese, Bengal) |
|
Naive |
| NEW! LiAb-VHHMAXTM |
VHH | Camel, llama, alpaca – 57 animals – Ideal for VHH generation | 1.51 X 1010 | Naive |
| LiAb-SFRabTM | scFv & Fab | Rabbit – 4 different breeds for maximized diversity | 1.09 X 1010 | Naive |
| LiPep-12 | Peptide 12-mer | / | 1.00 X 109 | Naive |
| LiPep-7 | Peptide 7-mer | / | 1.00 X 109 | Naive |
We have more libraries in scFv format because they are genetically more stable than the Fab format.
But the advantage of the Fab format is that it is possible to select in a more simple way the binders (by avoiding the risk of formation of dimers or trimers).
More libraries of other species may be available upon request.
A screening of the chosen library is carried out by ELISA in order to identify high-affinity binders of the protein of interest. During the interaction between the binders and the protein, binders will remain attached to the well while non-binders will be eliminated by washing.
Binders are eluted and then recovered to infect bacteria (with a helper phage) and increase their quantity. The repetition of this method is called « panning » (or also called « biopanning ») and allows to select the best binders, which are then isolated for validation in ELISA.
At the end, the DNA of the best binders is sequenced thereby allowing to produce them as recombinant proteins. ProteoGenix generally performs 3 to 5 steps of panning to ensure you get the best possible candidates.
ProteoGenix can generate high-diversity antibody libraries for phage display providing you with the most refined binders possible for your project. The construction of an immune library potentially increases the affinity of your final antibody against the targeted protein.
ProteoGenix is an expert in antibody production so many antibody libraries from various species can be generated at your convenience: Rabbit, Mouse, Sheep or even Alpaca and Llama (for VHH libraries).
Immunization.
Isolation of PBMC:
PCR amplification of antibodies:
cDNA VH/VL assembling construction of library:
Total:
Provides binders with a higher affinity compared to a naive library
More antibodies directed against the antigen of interest
Ideal for small and modified antigens (specific antibody with low capacity)
Allows work with any species as there is no fusion necessary compared to hybridoma technique
(example for rabbit monoclonal antibodies)
The library capacity is measured at 109 to 1016 different binders. We are able to offer you several types of libraries: scFv, Fab and VHH. The library screening is performed to identify binders with the best affinity.
Construction and screening of an immune library can be tailored to your research for high specificity and stability.
The screening process is based on our standard protocol of panning/biopanning and ProteoGenix can adapt this protocol to any request. You can find out more about the stages of biopanning in the screening from a naive library.
After 3 to 5 rounds of biopanning, ProteoGenix provides you with several binders (3 to 10) whilst giving you the option to ask for more if required.
At ProteoGenix, the choice of building and screening an immune library by phage display will allow you to produce monoclonal antibodies with the highest affinity for any species.
For more information or to get a quote for our phage display services with the use of immune libraries, don’t hesitate to contact us.
The creation of the first antibody libraries preceded the development of antibody display technologies. Their creation was made possible by the advance of molecular biology techniques and genomic studies. Today, it is possible to recover the antibody repertoire from any species and from hosts in any immune state (naïve/immune) to build high diversity and highly functional libraries adapted to display technologies. In this article, we explain how these libraries are generated and how they are used in antibody display. Check our frequently asked questions (FAQs) page about phage display for a complete overview of all steps of this robust process for antibody generation.
The onset of advanced molecular biology techniques created new possibilities to leverage the natural ingenuity of biological systems. In the late 1980s, this led to the construction of the first antibody libraries. These libraries were created by amplifying antibody encoding genes from lymphocytes, cloning them, and expressing them in bacterial systems.
These early attempts at transferring immune and naïve repertoires to the lab preceded the creation of antibody display techniques. At that time, the libraries would be either expressed as single-chain variable fragments (scFv) or as antibody combinatorial libraries of Fab fragments in bacteria. However, screening these libraries was quite labor-intensive and involved the use of radio-labeled antigens to identify the bacterial clones expressing antibodies with the highest affinity and specificity.
Shortly after these initial breakthroughs, the phage display technology was born. It created not only the possibility to screen libraries efficiently but also the ability to engineer antibodies by random and site-directed mutagenesis.
Genomic studies led to the discovery of conserved regions in antibody-encoding genes across different species. This discovery further prompted the development of primers targeting these regions, allowing the amplification of variable light and heavy chains (VL and VH, respectively) for many different host species, and subsequent cloning antibody-encoding genes using standard molecular biology techniques.
These VL and VH repertoires are commonly generated from naïve or immune hosts by harvesting either B lymphocytes (B cells) or peripheral blood mononuclear cells (PBMC) which consist of a mixture of T cells, B cells, and NK cells (natural killer cells), isolated from peripheral blood samples by simple density gradient centrifugation.
This initial harvesting step is followed by mRNA isolation, cDNA synthesis, and antibody gene amplification using a mixture of primers that ensures the highest antibody diversity is captured at this stage. These genes are subsequently cloned into the proper vector.
Phagemid vectors have become the most commonly used approach for antibody library generation. These vectors are typically derived from filamentous phage M13, a single-stranded DNA (ssDNA) phage with a circular genome capable of infecting strains of Escherichia coli harboring the F (fertility) pili. This phage carries all genes necessary for infection, replication, assembly, as well as all structural proteins. In contrast, phagemid vectors encode only a “minimal” version of the M13 phage containing key elements such as:
• a selection marker (i.e. antibiotic resistance gene)
• genes encoding for specific antibody fragments fused to one of the phage’s coat proteins
• the phage’s origin of replication – crucial for phage DNA replication
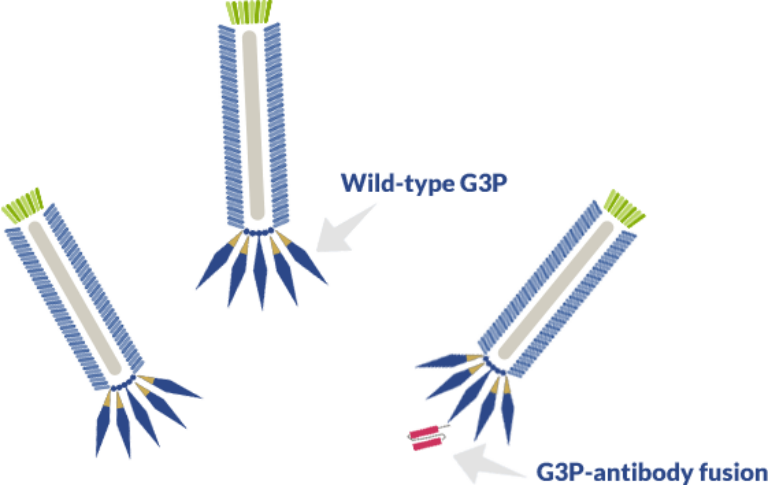
Antibody-encoding genes in the form of scFv (single-chain fragment variable regions), Fab, or VHH are typically fused to one of M13’s coat proteins – G3P, also known as pIII. About 4-5 copies of G3P are expressed on the tip of fully assembled M13 phages and these copies are responsible for binding to the F pili in E. coli.
However, fused copies of G3P cannot interact with E. coli’s F pili. Because, when all copies of the G3P protein are coupled to antibody fragments, the phage is unable to infect E. coli and, thus, no replication can take place.
The most common approach to circumventing this issue is the use phagemid vectors in conjugation with helper phages. Helpers contain all genes of the M13 phage essential for capsid production, assembly, replication, and budding, including the wild-type unfused GP3.
In E. coli, helper phage vectors compete with the phagemid encoding the G3P-antibody fusion protein. As a result, the majority of phage particles either have no antibody fragment displayed on its surface or display a single G3P-antibody fusion protein alongside wild-type G3P. The mixed phenotype allows the reconciliation of the phage’s role in antibody display with its ability to infect and replicate in E. coli.
The phagemid system also ensures that only a single antibody fragment is displayed per phage particle which, in turn, ensures only the binders with the highest affinity towards a specific antigen are enriched during the multiple rounds of biopanning.
Antibody libraries based on phagemid vectors have a critical advantage over full phage libraries – a higher transformation efficiency. Due to its “minimal” size, E. coli is more easily transformed by phagemid than by phage vectors. Moreover, since most resulting phage particles derived from phagemid libraries have only a single antibody fragment on its surface (monovalent), the selection of high-affinity antibodies is more efficient than when multivalent formats are used (phage vectors). In other words, the monovalent format ensures that antibodies with weak affinity can be easily washed off during panning campaigns.
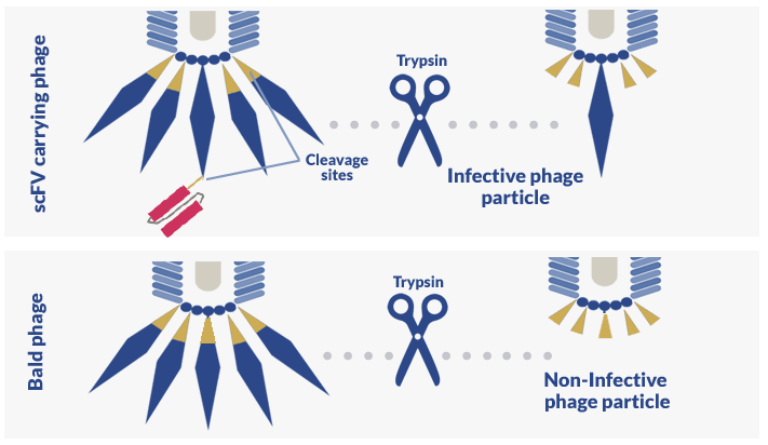
However, because phagemid libraries need to be amplified by direct competition with a helper phage with wild-type G3P, the vast majority of the phages will be devoid of antibody fragments (“bald” phages”) but still retain their infectivity.
To circumvent this issue and reduce the abundance of “bald” phages, a trypsin cleavage site is often introduced into the wild-type G3P. This modification ensures that, after panning and enrichment of high-affinity antibody fragments, trypsin treatment can be performed to render “bald” phages non-infective. The same trypsin cleavage site is also added to the linker region between the antibody fragment and G3P, so that antibody fragments can be cleaved from positive phages to allow them to infect E. coli.