Introduction
Imsidolimab Biosimilar, also known as Anti-IL36R mAb, is a monoclonal antibody that targets the interleukin-36 receptor (IL-36R). This therapeutic antibody has shown promising results in pre-clinical and clinical studies for the treatment of various inflammatory and autoimmune diseases. In this article, we will delve into the structure, activity, and potential applications of Imsidolimab Biosimilar.
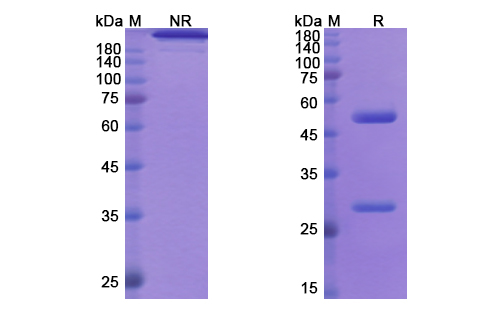
Structure of Imsidolimab Biosimilar
Imsidolimab Biosimilar is a fully humanized monoclonal antibody that is produced using recombinant DNA technology. It is composed of two heavy chains and two light chains, each with a molecular weight of approximately 25 kDa. The antibody has a Y-shaped structure, with two antigen-binding fragments (Fab) and one crystallizable fragment (Fc). The Fab region is responsible for binding to the IL-36R, while the Fc region is involved in effector functions such as antibody-dependent cell-mediated cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC).
Activity of Imsidolimab Biosimilar
Imsidolimab Biosimilar specifically targets the IL-36R, which is a receptor expressed on the surface of various immune cells, including T cells, dendritic cells, and macrophages. IL-36R is a member of the interleukin-1 (IL-1) receptor family and is involved in the regulation of inflammatory responses. When activated by its ligands, IL-36R triggers a cascade of signaling pathways that lead to the production of pro-inflammatory cytokines, such as interleukin-1 beta (IL-1β), interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-α).
By binding to the IL-36R, Imsidolimab Biosimilar blocks the interaction between the receptor and its ligands, thereby inhibiting the downstream signaling pathways and reducing the production of pro-inflammatory cytokines. This mechanism of action makes Imsidolimab Biosimilar a potent anti-inflammatory agent.
Potential Applications of Imsidolimab Biosimilar
Imsidolimab Biosimilar has shown promising results in pre-clinical and clinical studies for the treatment of various inflammatory and autoimmune diseases, including psoriasis, psoriatic arthritis, and hidradenitis suppurativa. These diseases are characterized by an overactive immune response, leading to chronic inflammation and tissue damage.
In a phase II clinical trial, Imsidolimab Biosimilar demonstrated significant efficacy in reducing the severity of psoriasis symptoms, such as skin lesions and itching, compared to placebo. The antibody also showed a favorable safety profile, with no serious adverse events reported. These results suggest that Imsidolimab Biosimilar could be a potential treatment option for patients with moderate to severe psoriasis.
Another potential application of Imsidolimab Biosimilar is in the treatment of psoriatic arthritis, a chronic inflammatory disease that affects the joints. In a phase II clinical trial, Imsidolimab Biosimilar showed significant improvement in joint symptoms, such as pain and swelling, in patients with psoriatic arthritis. The antibody also demonstrated a good safety profile, with no serious adverse events reported.
In addition to psoriasis and psoriatic arthritis, Imsidolimab Biosimilar has also shown promising results in the treatment of hidradenitis suppurativa, a chronic inflammatory skin disease. In a phase II clinical trial, the antibody significantly reduced the number of skin lesions and improved the quality of life in patients with hidradenitis suppurativa. The safety profile of Imsidolimab Biosimilar was also favorable, with no serious adverse events reported.
Conclusion
In summary, Imsidolimab Biosimilar is a promising therapeutic antibody that specifically targets the IL-36R and has shown significant efficacy in pre-clinical and clinical studies for the treatment of various inflammatory and autoimmune diseases. Its unique mechanism of action and favorable safety profile make it a potential treatment option for patients with psoriasis
There are no reviews yet.