Cart (0 Items)
Your cart is currently empty.
View ProductsIt looks like you are visiting from outside the EU. Switch to the US version to see local pricing in USD and local shipping.
Switch to US ($)The enzyme-linked immunosorbent assay (ELISA) is a popular plate-based method designed to detect, monitor, or quantify specific ligands in liquid samples such as peptides, proteins, antibodies, and hormones. Its popularity stems from flexibility in terms of format and easy parallelization, standardization, and miniaturization.
ELISA makes use of antigen-specific antibodies, an enzymatic tag (e.g. horseradish peroxidase tag, HRP), and a chromogenic substrate (e.g. 3,3′,5,5′-tetramethylbenzidine, TMB, for HRP), to measure specific ligands or interactions. There are currently four main types of ELISA tests:
Different ELISA kits have recently become available to monitor the COVID-19 pandemic. The unique advantages of the method in comparison to more conventional methods for research and diagnostics include the:
Unlike PCR-based tests, which are limited to the detection of viral particles, COVID-19 ELISA kits can be designed to detect the immune response to the SARS-CoV-2 virus, even after the pathogen has been eliminated from the patient’s organism.
This ability makes it an extremely useful tool in large-scale immune surveillance studies that allow monitoring the immune state of vast regions and populations, as well as, estimating the risks of development of new COVID-19 clusters.
Additionally, these tests are useful for determining the longevity and stability of the immune response induced by vaccine candidates currently undergoing preclinical and clinical trials.
Several COVID-19 disease markers have already been identified in the course of the pandemic. Molecules such as cytokines play a vital role in the onset of the exacerbated inflammatory response in the most severe cases of the COVID-19 disease.
Cytokines such as the interleukin-6 (IL-6), interleukin-10 (IL-10), tumor necrosis factor alpha (TNF-alpha), and interferon-γ induced protein 10 (IP-10), among others have been identified as particularly relevant indicators of disease severity. ELISA kits designed to detect these markers in patient’s serum and plasma should allow the fast diagnostics and quick adaptation of treatment strategies.
The COVID-19 pandemic has created an urgent need to develop fast and robust screening solutions. This need has, in turn, culminated in the development of specific ELISA kits that measure the interaction between important protein domains.
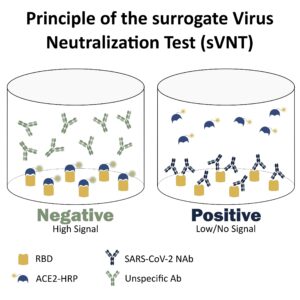
For instance, the recently developed surrogate Virus Neutralization Test (sVNT) is based on the principle of competitive ELISA, where an antibody with unknown activity competes with the human ACE2 receptor for the SARS-CoV-2 receptor-binding domain (RBD). This format allows the quick screening of an antibody’s potential for neutralizing the new coronavirus, as well as, provides a high-throughput solution for measuring and monitoring the neutralizing response in patients’ sera.