Introduction to Teplizumab Biosimilar – Anti-CD3 mAb – Research Grade
Teplizumab Biosimilar, also known as Anti-CD3 monoclonal antibody (mAb), is a promising therapeutic agent that has shown potential in the treatment of various autoimmune diseases. This biosimilar is a replica of the original Teplizumab, which is a humanized anti-CD3 mAb. The research grade of this biosimilar is specifically designed for use in laboratory research and pre-clinical studies. In this article, we will delve into the structure, activity, and applications of Teplizumab Biosimilar in detail.
Structure of Teplizumab Biosimilar
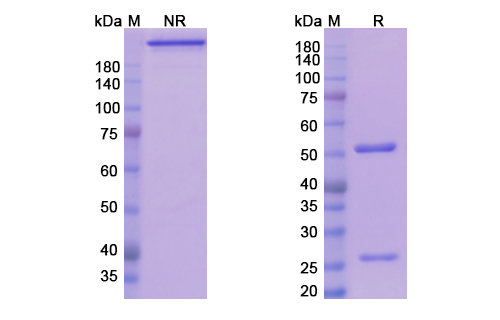
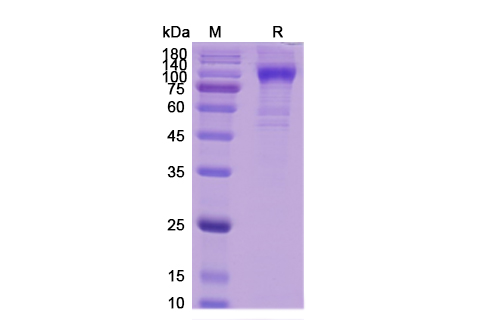
Teplizumab Biosimilar is a recombinant humanized IgG1 monoclonal antibody that is composed of two heavy and two light chains. The heavy chains are made up of four constant domains (CH1, CH2, CH3, and CH4) and one variable domain (VH), while the light chains consist of two constant domains (CL) and one variable domain (VL). The variable domains of both heavy and light chains are responsible for binding to the CD3 antigen, which is present on the surface of T cells.
The biosimilar is produced using recombinant DNA technology, where the gene for the variable region of the antibody is fused with the constant region of a human IgG1 antibody. This humanization process reduces the risk of immune reactions and increases the half-life of the antibody in the body.
Activity of Teplizumab Biosimilar
The main function of Teplizumab Biosimilar is to bind to the CD3 antigen on T cells and modulate their activity. CD3 is a complex protein that is involved in the activation and proliferation of T cells. By binding to CD3, Teplizumab Biosimilar inhibits the activation and proliferation of T cells, leading to a decrease in the production of inflammatory cytokines and a reduction in the activity of autoimmune diseases.
Teplizumab Biosimilar has also been shown to induce regulatory T cells (Tregs), which play a crucial role in maintaining immune tolerance and preventing autoimmune diseases. Tregs are responsible for suppressing the activity of other immune cells, including T cells, B cells, and dendritic cells. By increasing the number of Tregs, Teplizumab Biosimilar helps to restore immune balance and control autoimmune reactions.
Therapeutic Applications of Teplizumab Biosimilar
Teplizumab Biosimilar has shown promising results in the treatment of various autoimmune diseases, including type 1 diabetes, rheumatoid arthritis, and multiple sclerosis. In type 1 diabetes, the biosimilar has been shown to delay the progression of the disease and preserve beta-cell function, leading to improved glycemic control. In rheumatoid arthritis, Teplizumab Biosimilar has been found to reduce disease activity and improve symptoms. In multiple sclerosis, the biosimilar has shown potential in reducing the number and severity of relapses.
Moreover, Teplizumab Biosimilar has also been studied for its potential in preventing organ rejection in transplant patients. By modulating T cell activity, the biosimilar can help reduce the risk of graft rejection and improve transplant outcomes.
Conclusion
Teplizumab Biosimilar – Anti-CD3 mAb – Research Grade is a promising therapeutic agent with a unique mechanism of action. Its ability to bind to the CD3 antigen and modulate T cell activity makes it a potential treatment option for various autoimmune diseases. With further research and clinical trials, Teplizumab Biosimilar has the potential to improve the lives of patients suffering from these debilitating conditions.
Keywords: Teplizumab Biosimilar, Anti-CD3 mAb, research grade, antibody, therapeutic target
There are no reviews yet.