Introduction
Visilizumab Biosimilar, also known as Anti-CD3E monoclonal antibody (mAb), is a research grade antibody that has gained significant attention in the field of immunology. It is a biosimilar version of the therapeutic antibody, Visilizumab, which is used in the treatment of autoimmune diseases and transplant rejection. In this article, we will discuss the structure, activity, and potential applications of Visilizumab Biosimilar in scientific research.
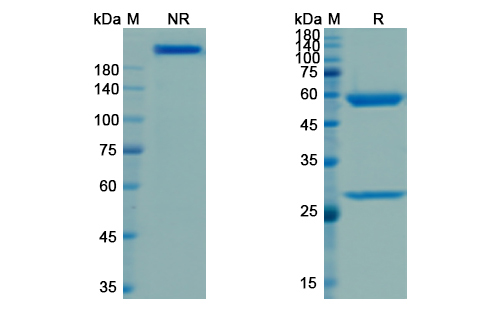
Structure of Visilizumab Biosimilar
Visilizumab Biosimilar is a recombinant humanized IgG1 monoclonal antibody that specifically targets the CD3E subunit of the T-cell receptor complex. It is composed of two heavy chains and two light chains, each containing a constant and a variable region. The variable region of Visilizumab Biosimilar is derived from a mouse anti-human CD3E antibody, while the constant region is humanized to reduce potential immunogenicity.
Mechanism of Action
Visilizumab Biosimilar binds to the CD3E subunit on the surface of T-cells, leading to the activation of these cells. This activation triggers a cascade of events, including the release of cytokines and the proliferation of T-cells. This mechanism is similar to that of the original Visilizumab, which is used in the treatment of autoimmune diseases and transplant rejection.
Activity of Visilizumab Biosimilar
Visilizumab Biosimilar has been shown to have potent immunomodulatory effects. It can activate T-cells and enhance their cytotoxicity, making it a potential therapeutic agent for the treatment of cancer. Additionally, Visilizumab Biosimilar has been found to have a regulatory effect on the immune system, making it a promising candidate for the treatment of autoimmune diseases.
Applications in Scientific Research
Visilizumab Biosimilar has been extensively studied in preclinical and clinical trials for its potential applications in scientific research. Some of the key areas where it has shown promising results include:
Cancer Immunotherapy The ability of Visilizumab Biosimilar to activate T-cells and enhance their cytotoxicity makes it a potential candidate for cancer immunotherapy. It has been studied in various types of cancer, including melanoma, colorectal cancer, and non-small cell lung cancer, and has shown promising results in preclinical studies.
Autoimmune Diseases
Visilizumab Biosimilar has shown potential in the treatment of autoimmune diseases such as type 1 diabetes, rheumatoid arthritis, and multiple sclerosis. Its immunomodulatory effects can help regulate the overactive immune response seen in these diseases.
Transplant Rejection
As a biosimilar of the original Visilizumab, Visilizumab Biosimilar has the potential to be used in transplant rejection. It can prevent the activation of T-cells that can lead to the rejection of transplanted organs.
Research Tools
Visilizumab Biosimilar can also be used as a research tool in various immunological studies. Its ability to specifically target the CD3E subunit of the T-cell receptor complex makes it a valuable tool for studying T-cell activation and proliferation.
Conclusion
In conclusion, Visilizumab Biosimilar is a promising research grade antibody with potential applications in cancer immunotherapy, autoimmune diseases, transplant rejection, and as a research tool. Its specific targeting of the CD3E subunit and potent immunomodulatory effects make it a valuable addition to the field of immunology research. Further studies and clinical trials are needed to fully explore the potential of Visilizumab Biosimilar in these areas.
There are no reviews yet.