Introduction to Ravulizumab Biosimilar – Anti-C5 mAb
Ravulizumab Biosimilar, also known as Anti-C5 mAb, is a research grade monoclonal antibody that targets the complement protein C5. This novel therapeutic agent has shown promising results in the treatment of various complement-mediated disorders, including paroxysmal nocturnal hemoglobinuria (PNH) and atypical hemolytic uremic syndrome (aHUS). In this article, we will discuss the structure, activity, and potential applications of Ravulizumab Biosimilar in detail.
Structure of Ravulizumab Biosimilar
Ravulizumab Biosimilar is a humanized monoclonal antibody that is derived from the parent antibody, eculizumab. It is composed of two identical heavy chains and two identical light chains, linked by disulfide bonds. The heavy chains consist of four constant domains (CH1, CH2, CH3, and CH4) and one variable domain (VH), while the light chains contain one constant domain (CL) and one variable domain (VL). The variable domains of both the heavy and light chains are responsible for binding to the target protein, C5.
Activity of Ravulizumab Biosimilar
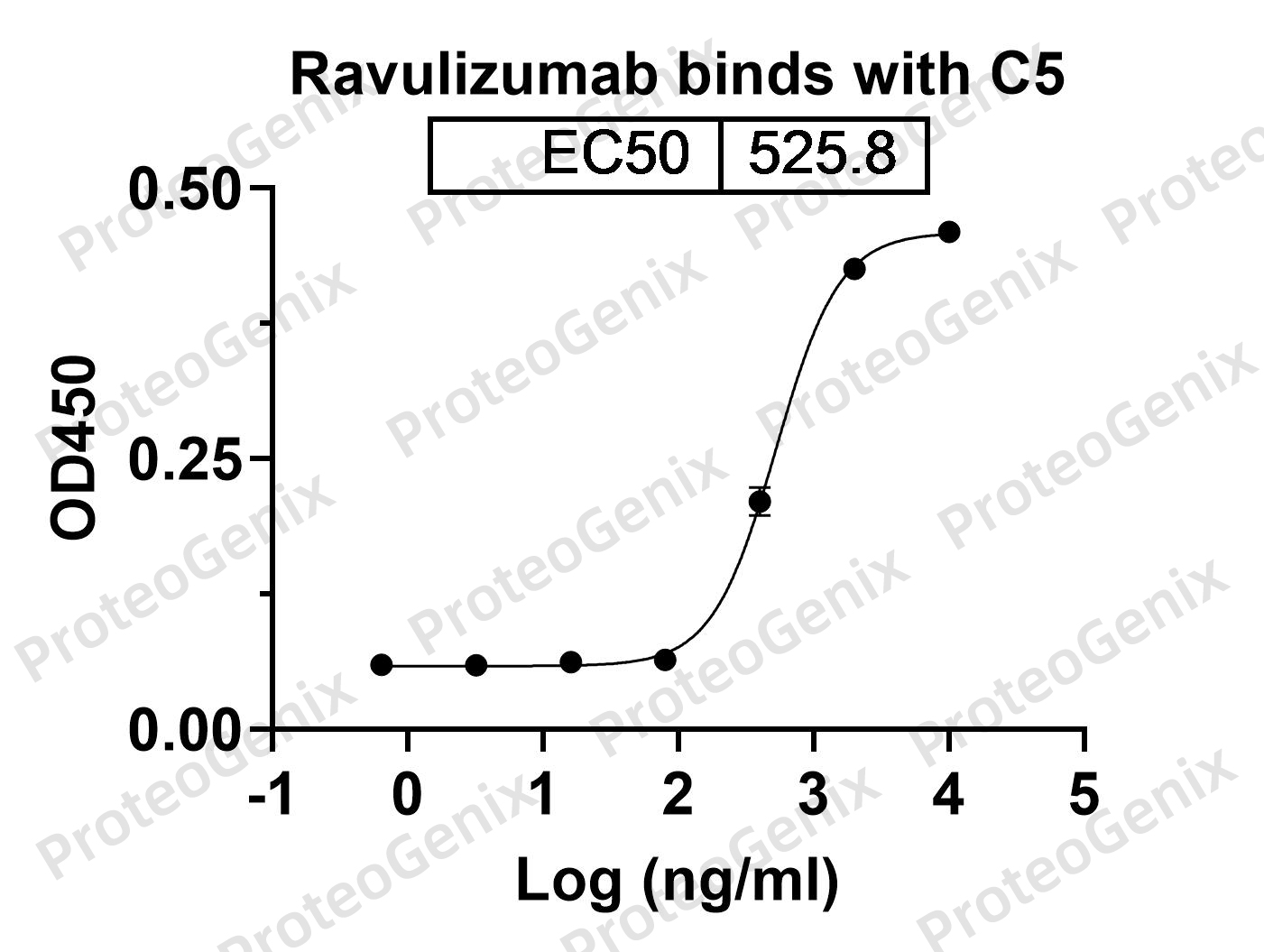
Ravulizumab Biosimilar exerts its therapeutic effect by specifically binding to the complement protein C5, thereby inhibiting its activity. C5 is a key component of the complement system, which plays a crucial role in the body’s immune response. When activated, C5 triggers a cascade of events that leads to inflammation and tissue damage. By blocking the activity of C5, Ravulizumab Biosimilar prevents the harmful effects of the complement system, making it an effective treatment for complement-mediated disorders.
Potential Applications of Ravulizumab Biosimilar
Ravulizumab Biosimilar has shown promising results in the treatment of PNH and aHUS, two rare and life-threatening disorders caused by abnormal activation of the complement system. PNH is a rare blood disorder characterized by the destruction of red blood cells, while aHUS is a rare kidney disorder that can lead to kidney failure. Both these disorders are caused by mutations in genes that regulate the complement system, resulting in uncontrolled activation of C5. By inhibiting C5, Ravulizumab Biosimilar can effectively treat these disorders and improve patient outcomes.
In addition to PNH and aHUS, Ravulizumab Biosimilar is also being investigated for its potential use in other complement-mediated disorders, such as myasthenia gravis, neuromyelitis optica spectrum disorder, and membranous nephropathy. These disorders are characterized by abnormal activation of the complement system, and Ravulizumab Biosimilar has shown promising results in preclinical studies and early clinical trials.
Conclusion
In summary, Ravulizumab Biosimilar is a research grade monoclonal antibody that specifically targets the complement protein C5. Its unique structure and mechanism of action make it a promising therapeutic agent for the treatment of various complement-mediated disorders. With ongoing research and clinical trials, Ravulizumab Biosimilar has the potential to improve the lives of patients suffering from these rare and debilitating disorders.
There are no reviews yet.