Introduction
Omodenbamab Biosimilar, also known as Anti-SpA mAb, is a monoclonal antibody that has been developed as a potential therapeutic agent for the treatment of spondyloarthritis (SpA). This biosimilar is a highly specific and potent antibody that targets a key protein involved in the pathogenesis of SpA. In this article, we will provide a comprehensive scientific description of Omodenbamab Biosimilar, including its structure, activity, and potential applications.
Structure of Omodenbamab Biosimilar
Omodenbamab Biosimilar is a recombinant humanized monoclonal antibody that is derived from the parent antibody, Omodenbamab. It is composed of two identical heavy chains and two identical light chains, each with a molecular weight of approximately 150 kDa. The antibody has a Y-shaped structure, with two antigen-binding fragments (Fab) and one crystallizable fragment (Fc). The Fab regions are responsible for binding to the target protein, while the Fc region is involved in immune effector functions.
Activity of Omodenbamab Biosimilar
Omodenbamab Biosimilar specifically targets the protein interleukin-23 (IL-23), which is a key cytokine involved in the pathogenesis of SpA. IL-23 is known to promote the differentiation and activation of Th17 cells, which play a critical role in the development of SpA. By binding to IL-23, Omodenbamab Biosimilar inhibits its activity and prevents the differentiation and activation of Th17 cells.
In addition, Omodenbamab Biosimilar also has potent anti-inflammatory effects. It has been shown to suppress the production of pro-inflammatory cytokines, such as IL-6 and TNF-alpha, which are also involved in the pathogenesis of SpA. This anti-inflammatory activity helps to reduce the symptoms of SpA, such as joint pain and swelling, and improve overall disease outcomes.
Potential Applications of Omodenbamab Biosimilar
Omodenbamab Biosimilar has shown promising results in preclinical and clinical studies as a potential treatment for SpA. It has been demonstrated to be safe and well-tolerated in human subjects, with no significant adverse effects reported. In addition, it has shown efficacy in reducing disease activity and improving symptoms in patients with SpA.
The potential applications of Omodenbamab Biosimilar extend beyond SpA. IL-23 is also involved in the pathogenesis of other autoimmune diseases, such as psoriasis and inflammatory bowel disease. Therefore, Omodenbamab Biosimilar may have potential applications in these conditions as well. Further research and clinical trials are needed to explore these potential uses.
Conclusion
In summary, Omodenbamab Biosimilar is a recombinant humanized monoclonal antibody that specifically targets IL-23, a key cytokine involved in the pathogenesis of SpA. It has potent anti-inflammatory effects and has shown promising results in preclinical and clinical studies as a potential treatment for SpA. With its unique structure and activity, Omodenbamab Biosimilar has the potential to improve the lives of patients with SpA and other autoimmune diseases. Further research and clinical trials are needed to fully understand its therapeutic potential.
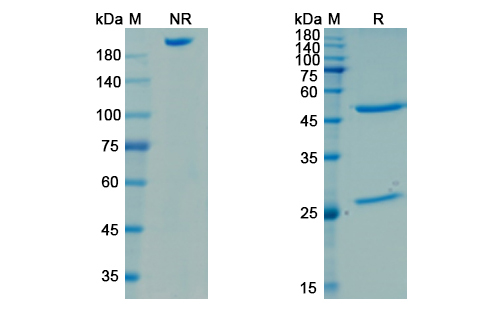
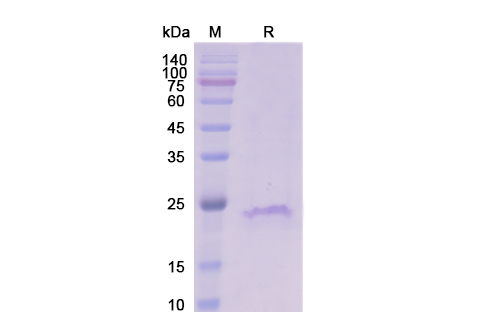
There are no reviews yet.