Bavunalimab Biosimilar – Anti-LAG3,CTLA4 mAb – Research Grade
Bavunalimab Biosimilar – Anti-LAG3,CTLA4 mAb – Research Grade: A Novel Antibody Targeting Immune Checkpoint Pathways
Introduction
Bavunalimab Biosimilar is a novel antibody that targets two important immune checkpoint pathways – LAG3 and CTLA4. This research grade antibody has shown promising results in preclinical studies and is currently being evaluated in clinical trials for its potential as a therapeutic agent in various cancers.
Structure of Bavunalimab Biosimilar
Bavunalimab Biosimilar is a monoclonal antibody that is produced using recombinant DNA technology. It is a chimeric antibody, meaning it is composed of both human and mouse components. The variable region of the antibody is derived from a mouse antibody, while the constant region is of human origin. This structure allows for better binding to the target molecules and reduces the risk of immune reactions in patients.
Mechanism of Action
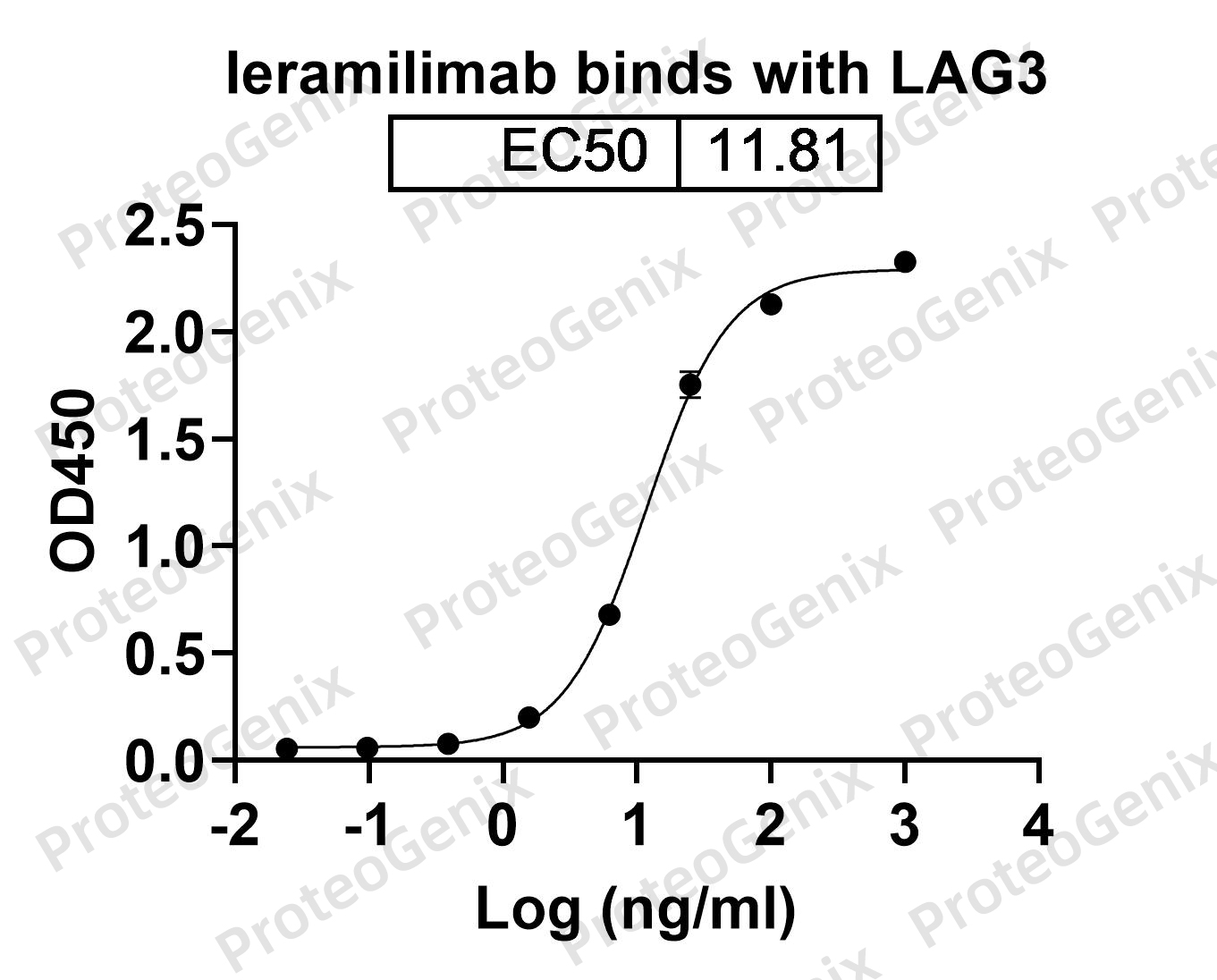
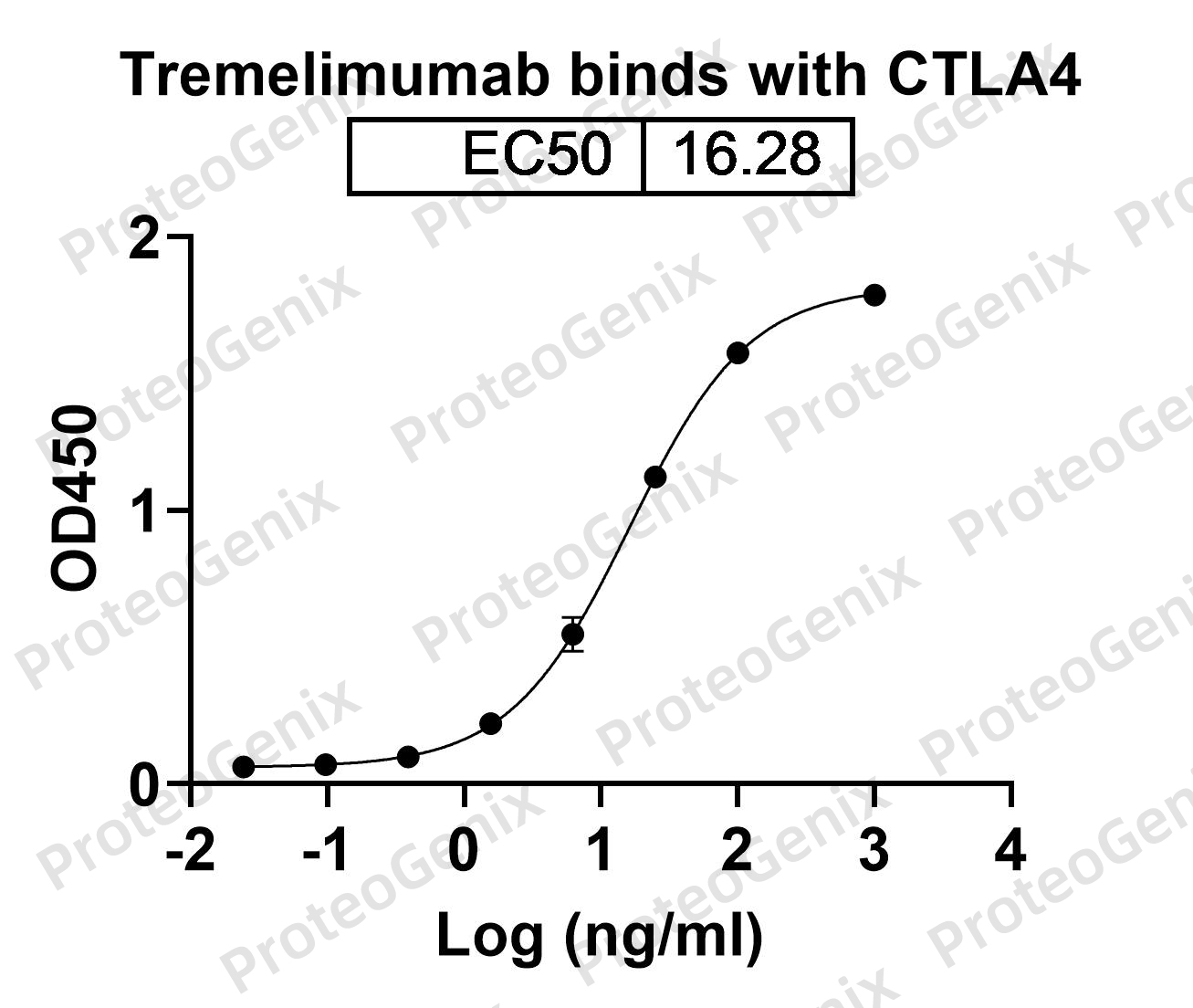
Bavunalimab Biosimilar works by targeting two important immune checkpoint pathways – LAG3 and CTLA4. These pathways play a crucial role in regulating the immune response and preventing excessive activation of immune cells. However, cancer cells can hijack these pathways to evade detection and destruction by the immune system. Bavunalimab Biosimilar blocks the interaction between LAG3 and its ligand, as well as the interaction between CTLA4 and its ligand, thereby restoring the immune response against cancer cells.
Applications of Bavunalimab Biosimilar
As an antibody targeting immune checkpoint pathways, Bavunalimab Biosimilar has potential applications in various cancers. It has shown promising results in preclinical studies for the treatment of melanoma, lung cancer, and colorectal cancer. It is also being evaluated in clinical trials for its efficacy in combination with other therapies, such as chemotherapy and other immunotherapies.
Research Grade Antibody
Bavunalimab Biosimilar is currently in the research grade stage, meaning it is being studied in preclinical and clinical trials to determine its safety and efficacy. It is not yet approved for clinical use, but the results from these studies will pave the way for its potential as a therapeutic agent in the future.
Conclusion
Bavunalimab Biosimilar is a novel antibody that targets two important immune checkpoint pathways – LAG3 and CTLA4. Its unique structure and mechanism of action make it a promising candidate for the treatment of various cancers. As a research grade antibody, it is currently being evaluated in clinical trials and has the potential to become an important therapeutic agent in the future.
Keywords:
Antibody, Bavunalimab Biosimilar, Anti-LAG3, CTLA4 mAb, immune checkpoint pathways, therapeutic target, cancer, preclinical studies, clinical trials, research grade.

There are no reviews yet.