Introduction to Vunakizumab Biosimilar – Anti-IL17A mAb
Vunakizumab Biosimilar – Anti-IL17A mAb is a research grade monoclonal antibody (mAb) that targets the cytokine interleukin-17A (IL-17A). This biosimilar is designed to mimic the structure and function of the original Vunakizumab, a fully human mAb developed by Merck for the treatment of autoimmune diseases. In this article, we will delve into the structure, activity, and potential applications of this promising biosimilar.
Structure of Vunakizumab Biosimilar – Anti-IL17A mAb
Vunakizumab Biosimilar – Anti-IL17A mAb is a recombinant, humanized mAb that is produced in a mammalian cell expression system. It consists of two identical heavy chains and two identical light chains, each with a molecular weight of approximately 150 kDa. The heavy chains are composed of four constant domains (CH1, CH2, CH3, and CH4) and one variable domain (VH), while the light chains have two constant domains (CL and CL’) and one variable domain (VL). The VH and VL domains come together to form the antigen-binding site, which is responsible for the specificity of the mAb.
Activity of Vunakizumab Biosimilar – Anti-IL17A mAb
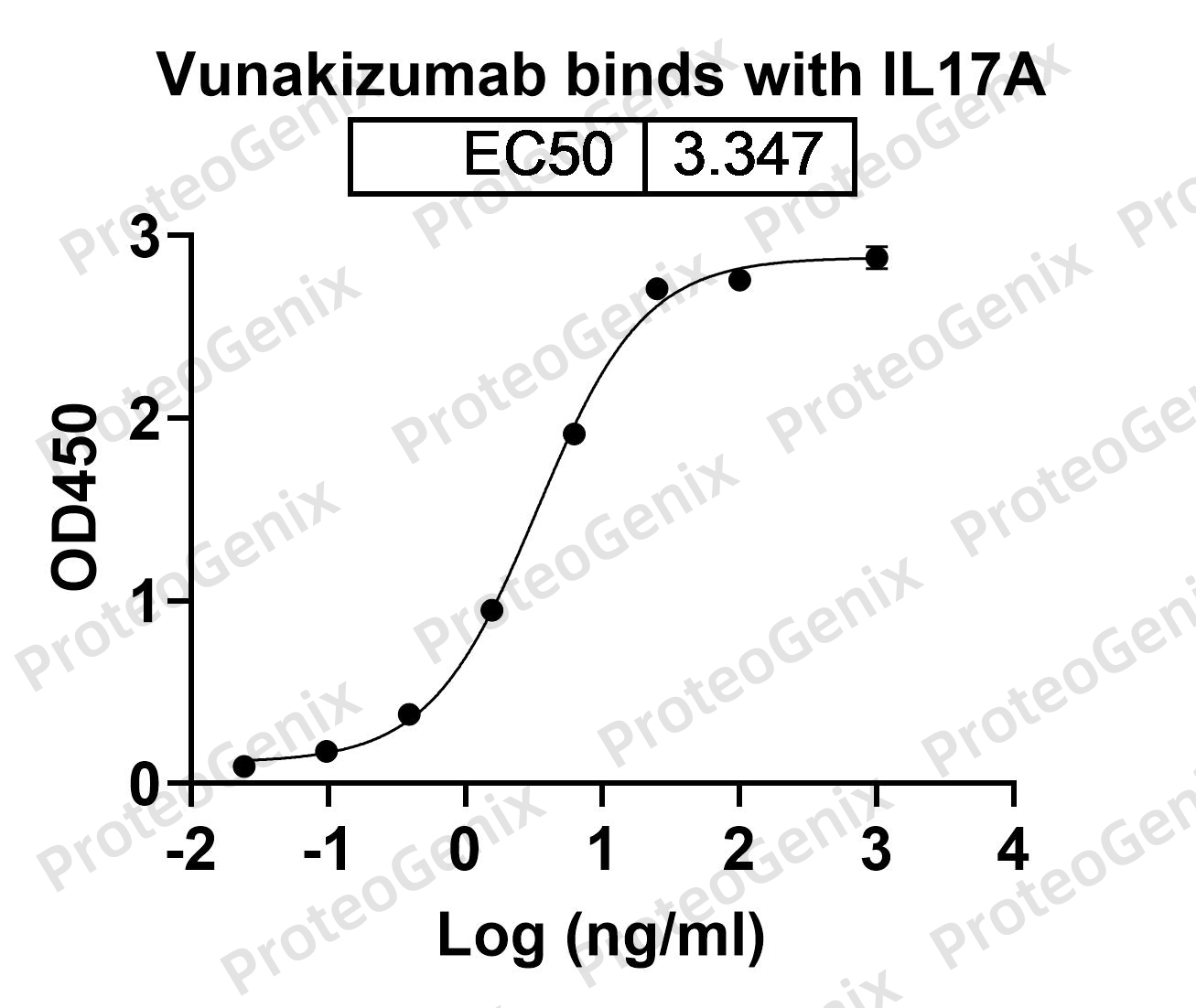
IL-17A is a pro-inflammatory cytokine that plays a crucial role in the pathogenesis of autoimmune diseases such as psoriasis, rheumatoid arthritis, and inflammatory bowel disease. Vunakizumab Biosimilar – Anti-IL17A mAb binds specifically to IL-17A, preventing it from binding to its receptor and thereby inhibiting its activity. This leads to a decrease in the production of other pro-inflammatory cytokines and chemokines, ultimately reducing inflammation and tissue damage.
Applications of Vunakizumab Biosimilar – Anti-IL17A mAb
Due to its ability to target IL-17A, Vunakizumab Biosimilar – Anti-IL17A mAb has potential applications in the treatment of various autoimmune diseases. It has been shown to be effective in clinical trials for psoriasis, with a significant reduction in disease severity and improvement in symptoms. In addition, it has also shown promising results in clinical trials for rheumatoid arthritis and ankylosing spondylitis.
Furthermore, Vunakizumab Biosimilar – Anti-IL17A mAb has been investigated for its potential in treating other autoimmune diseases such as psoriatic arthritis, Crohn’s disease, and ulcerative colitis. These diseases share a common pathogenesis involving IL-17A, making Vunakizumab Biosimilar – Anti-IL17A mAb a promising therapeutic option.
Conclusion
In summary, Vunakizumab Biosimilar – Anti-IL17A mAb is a research grade mAb that targets IL-17A, a key cytokine involved in the pathogenesis of various autoimmune diseases. Its structure and activity make it a promising therapeutic option for conditions such as psoriasis, rheumatoid arthritis, and inflammatory bowel disease. With ongoing research and clinical trials, Vunakizumab Biosimilar – Anti-IL17A mAb has the potential to provide new treatment options for patients suffering from these debilitating diseases.
There are no reviews yet.