Introduction
Vibecotamab Biosimilar, also known as Anti-CD3 epsilon, IL3RA mAb, is a monoclonal antibody that has been developed as a biosimilar to the therapeutic antibody, Vibecotamab. This biosimilar is designed to target the CD3 epsilon subunit of the IL3RA receptor, making it a promising therapeutic option for a variety of diseases.
Structure
Vibecotamab Biosimilar is a fully humanized IgG1 monoclonal antibody, which means it is derived from human cells and has a structure similar to natural human antibodies. It is composed of two heavy chains and two light chains, connected by disulfide bonds. The antibody has a molecular weight of approximately 150 kDa and a half-life of around 21 days in the human body.
Activity
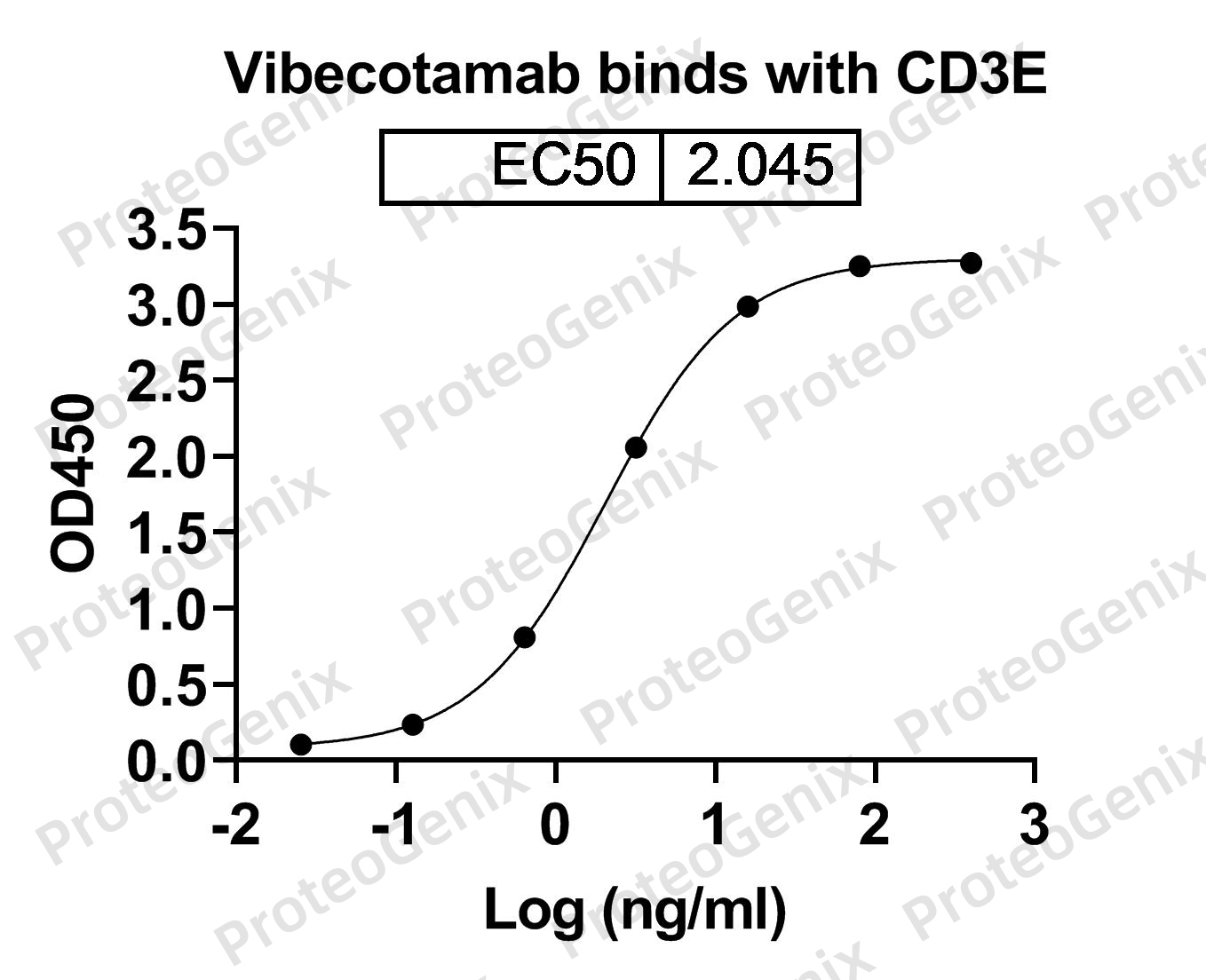
The main function of Vibecotamab Biosimilar is to bind to the CD3 epsilon subunit of the IL3RA receptor. This subunit is found on the surface of T cells, which are a type of white blood cell involved in the immune response. By binding to the CD3 epsilon subunit, Vibecotamab Biosimilar activates T cells and stimulates their proliferation, leading to an enhanced immune response.
Application
Vibecotamab Biosimilar has shown promising results in preclinical studies for the treatment of various diseases. Its main application is in the field of oncology, where it has been studied as a potential treatment for various types of cancer, including solid tumors and hematological malignancies. The antibody has also shown potential in the treatment of autoimmune diseases, such as rheumatoid arthritis and multiple sclerosis.
In oncology, Vibecotamab Biosimilar has been shown to induce tumor cell death through the activation of T cells. This mechanism of action makes it a promising option for the treatment of solid tumors, which are often difficult to target with traditional therapies. In addition, the antibody has been shown to have a synergistic effect when combined with other cancer treatments, such as chemotherapy and radiation therapy.
In autoimmune diseases, Vibecotamab Biosimilar has been shown to modulate the immune response, leading to a reduction in inflammation and disease symptoms. This makes it a potential treatment option for patients who do not respond well to traditional therapies or experience significant side effects.
Research Grade
Vibecotamab Biosimilar is currently in the research grade stage, which means it is still undergoing preclinical and clinical studies to determine its safety and efficacy in humans. These studies are necessary before the antibody can be approved for use in patients.
Conclusion
In summary, Vibecotamab Biosimilar is a promising monoclonal antibody that targets the CD3 epsilon subunit of the IL3RA receptor. Its structure, activity, and potential applications make it a promising therapeutic option for a variety of diseases, particularly in the fields of oncology and autoimmune diseases. With continued research and development, this biosimilar has the potential to improve patient outcomes and contribute to the advancement of medical treatments.
There are no reviews yet.