Introduction
Nerelimomab Biosimilar, also known as Anti-TNFSF2 or TNF-alpha monoclonal antibody (mAb), is a research grade therapeutic agent that targets the cytokine TNF-alpha. This protein is a key mediator of inflammation and is involved in a variety of diseases such as rheumatoid arthritis, Crohn’s disease, and psoriasis. Nerelimomab Biosimilar is a promising treatment option for these conditions and has the potential to improve patient outcomes.
Structure of Nerelimomab Biosimilar
Nerelimomab Biosimilar is a monoclonal antibody, meaning it is a laboratory-produced protein that mimics the body’s natural antibodies. The antibody is composed of two heavy chains and two light chains, each containing a variable region and a constant region. The variable region is responsible for binding to the target protein, while the constant region is involved in immune system activation.
Activity of Nerelimomab Biosimilar
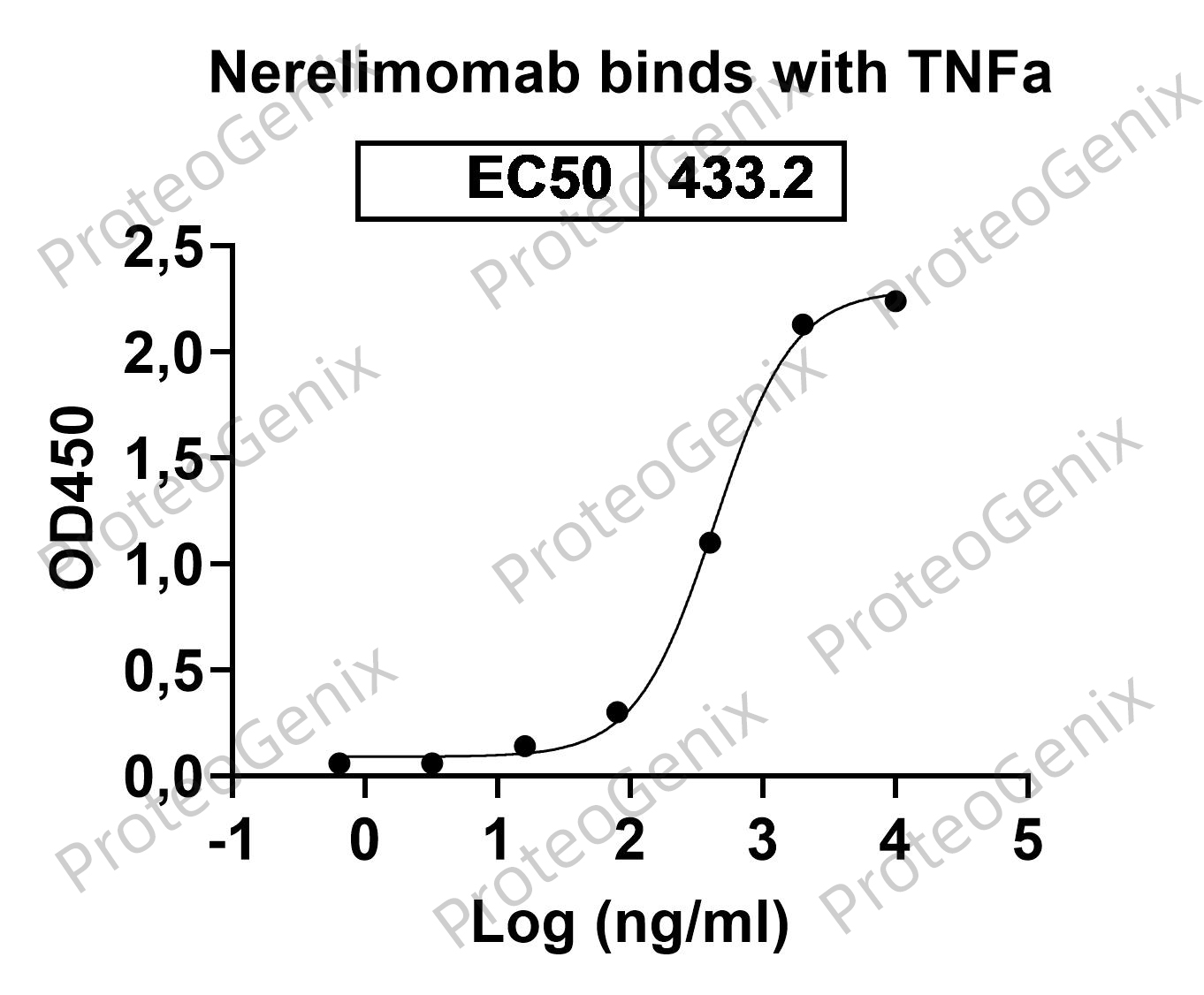
Nerelimomab Biosimilar binds specifically to TNF-alpha, preventing it from binding to its receptors and exerting its pro-inflammatory effects. This leads to a decrease in inflammation and symptoms associated with diseases where TNF-alpha is overproduced. Additionally, Nerelimomab Biosimilar can also promote the clearance of TNF-alpha by activating the immune system to recognize and remove the bound protein.
Application of Nerelimomab Biosimilar
Nerelimomab Biosimilar has shown promising results in preclinical studies for the treatment of various inflammatory diseases. In a mouse model of rheumatoid arthritis, Nerelimomab Biosimilar significantly reduced joint inflammation and destruction. It has also been shown to be effective in treating inflammatory bowel disease, with a reduction in symptoms and improvement in intestinal tissue damage.
In addition to its potential as a therapeutic agent, Nerelimomab Biosimilar has also been used in research to study the role of TNF-alpha in various diseases. By blocking TNF-alpha activity, researchers can better understand the mechanisms of inflammation and potentially identify new therapeutic targets.
Future Directions
Nerelimomab Biosimilar is currently in the research and development stage, with clinical trials underway to evaluate its safety and efficacy in human patients. If successful, it has the potential to become a valuable treatment option for inflammatory diseases with limited treatment options. Furthermore, ongoing research on the role of TNF-alpha in other diseases may lead to the discovery of new applications for Nerelimomab Biosimilar.
Conclusion
Nerelimomab Biosimilar is a promising research grade therapeutic agent that targets the pro-inflammatory cytokine TNF-alpha. Its specific binding to TNF-alpha and ability to promote its clearance make it a potential treatment option for various inflammatory diseases. Ongoing research and clinical trials will provide more insight into the efficacy and safety of this antibody, paving the way for its potential use in the clinic.
There are no reviews yet.