Introduction
Cibisatamab Biosimilar, also known as Anti-CEACAM5&CD3E,CD3E mAb, is a monoclonal antibody used for therapeutic purposes. It is a biosimilar version of the original Cibisatamab, which is a fully humanized monoclonal antibody targeting CEACAM5 and CD3E antigens. This biosimilar version is produced through recombinant DNA technology and has shown promising results in preclinical and clinical studies. In this article, we will discuss the structure, activity, and potential applications of Cibisatamab Biosimilar.
Structure of Cibisatamab Biosimilar
Cibisatamab Biosimilar is a chimeric monoclonal antibody, meaning it is composed of both human and non-human components. It is made up of two heavy chains and two light chains, which are connected by disulfide bonds. The heavy chains are approximately 50 kDa in size, while the light chains are around 25 kDa. The antibody has a molecular weight of approximately 150 kDa.
The variable regions of Cibisatamab Biosimilar are derived from a mouse monoclonal antibody, while the constant regions are from a human immunoglobulin G1 (IgG1) antibody. This structure allows for high specificity and affinity towards its target antigens.
Activity of Cibisatamab Biosimilar
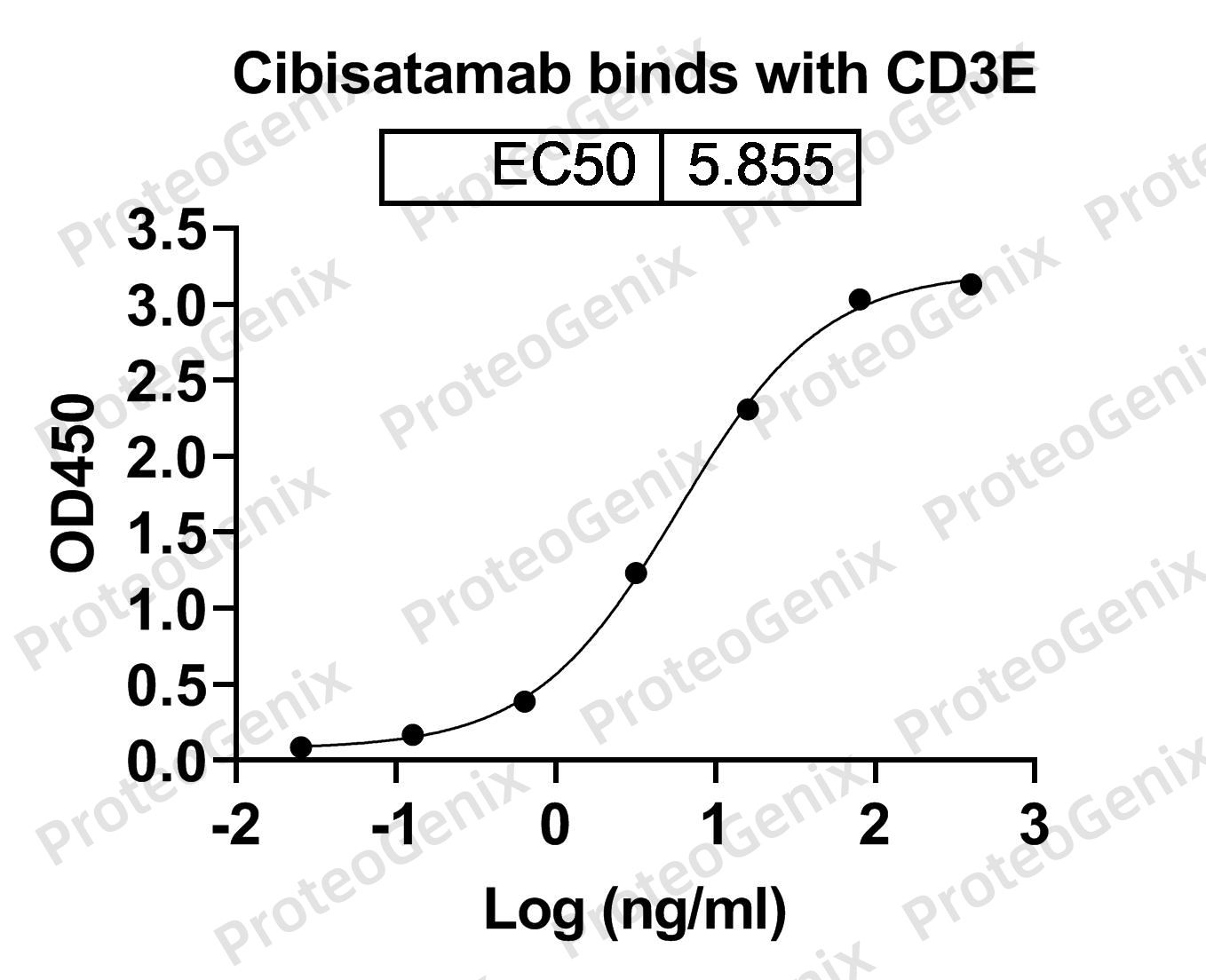
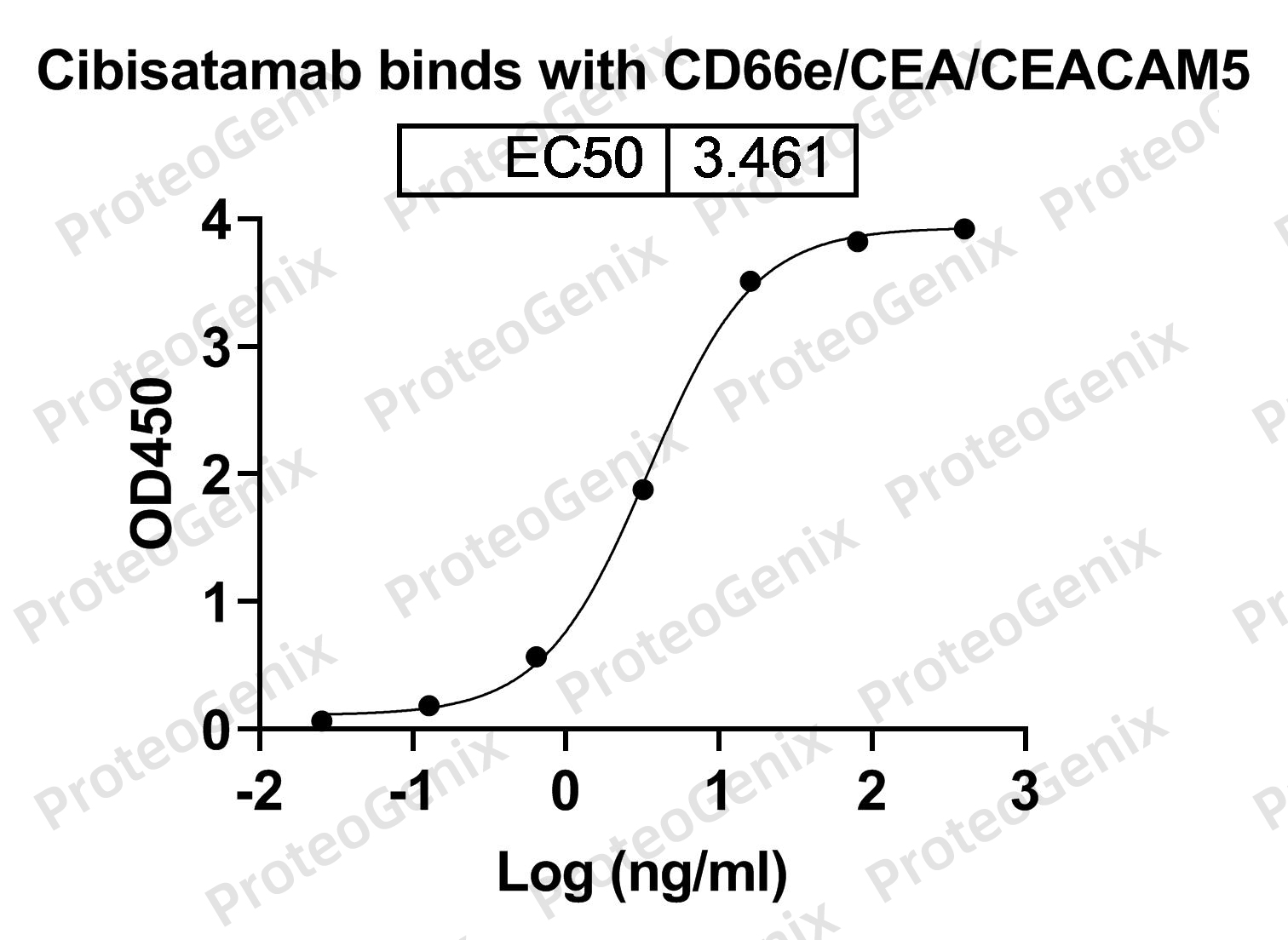
Cibisatamab Biosimilar works by binding to two different antigens, CEACAM5 and CD3E, on the surface of cancer cells. CEACAM5 is a glycoprotein that is overexpressed in many types of cancer, including colorectal, lung, and breast cancer. CD3E is a protein found on the surface of T cells, a type of immune cell. By targeting both antigens, Cibisatamab Biosimilar can activate T cells to attack and kill cancer cells.
Once bound to its target antigens, Cibisatamab Biosimilar triggers a cascade of immune responses, including antibody-dependent cellular cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC). These mechanisms work by recruiting other immune cells and proteins to destroy the targeted cancer cells.
Potential Applications of Cibisatamab Biosimilar
Cibisatamab Biosimilar is currently being investigated for its potential use in the treatment of various types of cancer. Preclinical studies have shown promising results in colorectal, lung, and breast cancer models. In a phase 1 clinical trial, Cibisatamab Biosimilar showed promising results in patients with advanced solid tumors, including those with colorectal cancer.
In addition to its potential as a monotherapy, Cibisatamab Biosimilar is also being studied in combination with other cancer treatments. For example, it is being investigated in combination with chemotherapy for the treatment of colorectal cancer, and with immune checkpoint inhibitors for the treatment of lung cancer.
Conclusion
In conclusion, Cibisatamab Biosimilar is a chimeric monoclonal antibody that targets CEACAM5 and CD3E antigens on the surface of cancer cells. Its unique structure and mechanism of action make it a promising therapeutic option for various types of cancer. Further research and clinical trials are needed to fully understand the potential of this biosimilar in cancer treatment.
There are no reviews yet.