General information on Tocilizumab Therapeutic Antibody
The tocilizumab antibody is an IgG1 kappa monoclonal protein obtained from mouse hybridoma and subsequently humanized for use in antibody therapy. Tocilizumab acts as an immunosuppressant and it has been designed to bind the human interleukin 6 receptor (IL-6R), which occurs in membrane-bound or soluble form. The receptor is known to bind IL-6, a 21-26 kDa glycoprotein, and mediate proinflammatory reactions in response to infections or tissue injuries.
IL-6 production is triggered locally and only during the early stages of inflammation. Despite its key role in the immune response, IL-6 overproduction has been observed in different chronic inflammatory diseases and some forms of cancer. Moreover, its presence has been linked with the excessive and potentially fatal inflammatory responses known as “cytokine storm.”
Tocilizumab is one of the first humanized monoclonal antibodies ever to be licensed and commercialized as an efficient IL6R blocker. Due to its specificity, it is recurrently used to treat autoimmune conditions such as rheumatoid arthritis, cytokine release syndrome (or “cytokine storm”), and polyarticular juvenile idiopathic arthritis; some forms of cancer such as Castleman’s disease; and inflammatory diseases such as giant cell arteritis.
Recent studies showed that IL-6 levels in COVID-19 patients on ventilators can be 10 times higher in comparison to patients with milder symptoms of the disease. Exacerbated levels of the proinflammatory molecule have previously been observed in other coronavirus-mediated diseases such as the Severe Acute Respiratory Syndrome (SARS) and Middle East Respiratory Syndrome (MERS). In these diseases as in the case of COVID-19, the leading cause of death appears to be severe lung inflammation in the form of “cytokine storm.”
The efficiency of Tocilizumab in the COVID-19 disease has been recently reported in several observational studies, including a study from Italy where researchers reported improved oxygenation in 74% and 65% of the patients on mechanical or non-invasive ventilation, respectively. Multiple randomized, double-blind, and placebo-controlled clinical trials are currently underway to validate the efficiency of this IgG1 antibody for the treatment of the COVID-19 disease. This product is for research use only.
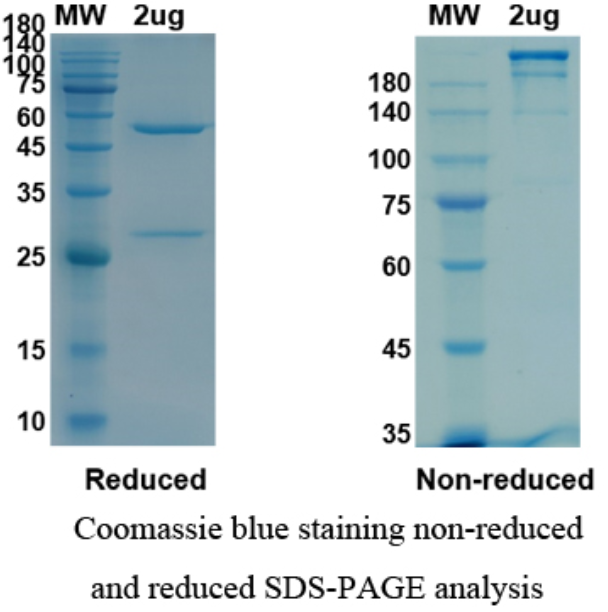
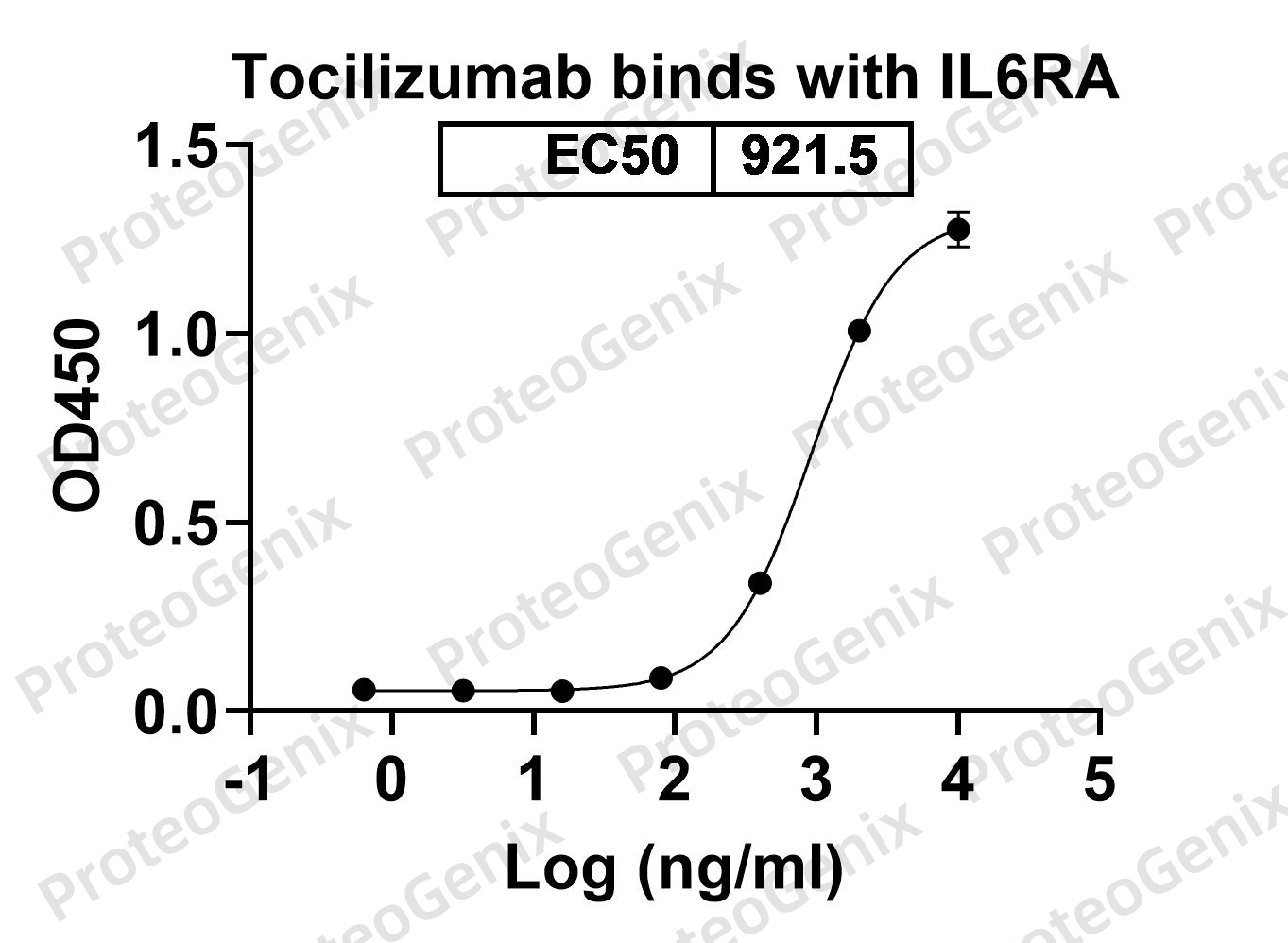
There are no reviews yet.