Introduction to Sintilimab Biosimilar – Anti-PDCD1, PD1, CD279 mAb – Research Grade Sintilimab Biosimilar, also known as Anti-PDCD1, PD1, CD279 mAb, is a monoclonal antibody that targets the programmed cell death protein 1 (PD-1) receptor. It is a biosimilar of the original drug, Sintilimab, which was developed by the Chinese biopharmaceutical company Innovent Biologics. This biosimilar is a research grade version of the drug, meaning it is intended for use in laboratory research and not for clinical use.
Structure of Sintilimab Biosimilar
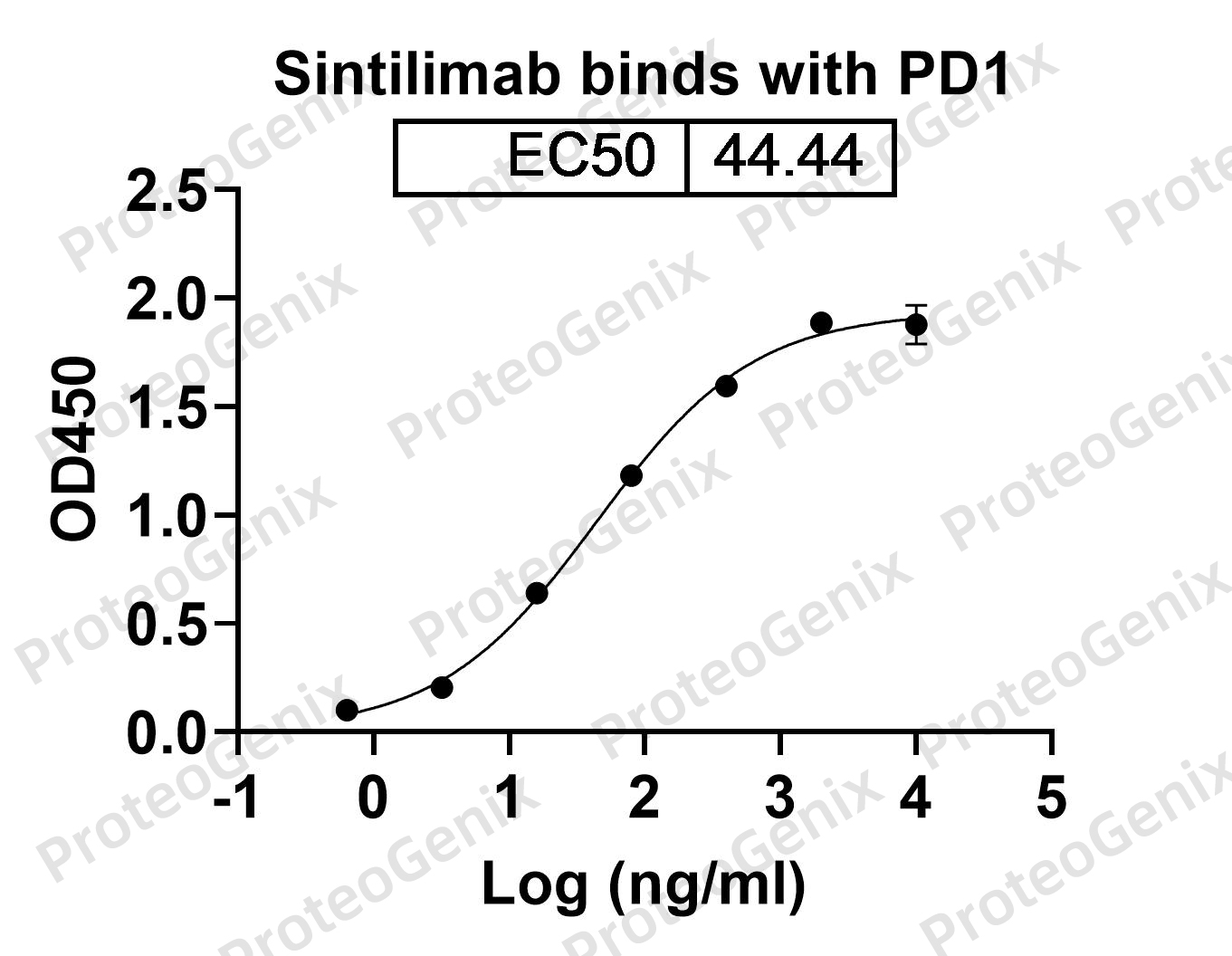
Sintilimab Biosimilar is a fully humanized monoclonal antibody, meaning it is derived from human cells and has a structure similar to the body’s own antibodies. It is composed of two heavy chains and two light chains, which are connected by disulfide bonds. The antibody has a molecular weight of approximately 150 kDa.
Mechanism of Action
Sintilimab Biosimilar works by binding to the PD-1 receptor on the surface of T cells, a type of immune cell. The PD-1 receptor is responsible for regulating the immune response by preventing T cells from attacking normal cells in the body. However, in cancer, the PD-1 receptor can be hijacked by tumor cells to evade the immune system. Sintilimab Biosimilar blocks the interaction between PD-1 and its ligands, PD-L1 and PD-L2, thereby allowing the immune system to recognize and attack cancer cells.
Applications of Sintilimab Biosimilar
Sintilimab Biosimilar has shown promising results in preclinical studies and is currently being evaluated in clinical trials for the treatment of various types of cancer, including non-small cell lung cancer, hepatocellular carcinoma, and esophageal squamous cell carcinoma. It is also being studied in combination with other cancer therapies, such as chemotherapy and other immunotherapies.
Non-Small Cell Lung Cancer (NSCLC) NSCLC is the most common type of lung
cancer, accounting for approximately 85% of all cases. Sintilimab Biosimilar has shown significant antitumor activity in preclinical studies and has been approved in China for the treatment of advanced or metastatic NSCLC. It is currently being evaluated in clinical trials for the treatment of NSCLC in other countries.
Hepatocellular Carcinoma (HCC) HCC is the most common type of liver
cancer and is often diagnosed at an advanced stage. Sintilimab Biosimilar has shown promising results in preclinical studies and is currently being evaluated in clinical trials for the treatment of advanced HCC. It has also been approved in China for the treatment of advanced HCC.
Esophageal Squamous Cell Carcinoma (ESCC)
ESCC is a type of cancer that affects the esophagus, the tube that connects the throat to the stomach. Sintilimab Biosimilar has shown significant antitumor activity in preclinical studies and is currently being evaluated in clinical trials for the treatment of ESCC. It has also been approved in China for the treatment of advanced ESCC.
Conclusion
Sintilimab Biosimilar is a research grade version of the original drug, Sintilimab, and is a promising therapeutic option for the treatment of various types of cancer. Its mechanism of action, targeting the PD-1 receptor, has shown significant antitumor activity in preclinical studies and is currently being evaluated in clinical trials. With its potential to improve outcomes for cancer patients, Sintilimab Biosimilar is a valuable addition to the arsenal of cancer therapies.
There are no reviews yet.