Introduction
Runimotamab Biosimilar, also known as Anti-ERBB2,CD3E mAb, is a monoclonal antibody that has been developed as a biosimilar to the original drug, trastuzumab. It is a targeted therapy that specifically binds to the HER2 receptor (ERBB2) and the CD3E protein on the surface of cancer cells. This article will provide a scientific description of the structure, activity, and application of Runimotamab Biosimilar in research grade.
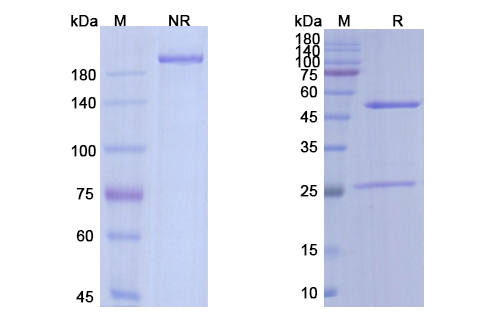
Structure of Runimotamab Biosimilar
Runimotamab Biosimilar is a recombinant humanized monoclonal antibody, meaning it is produced in a laboratory using human DNA sequences. It is composed of two heavy chains and two light chains, connected by disulfide bonds. The heavy chains are approximately 50 kDa in size, while the light chains are about 25 kDa. The antibody has a molecular weight of approximately 150 kDa.
The variable regions of the antibody, responsible for binding to the target proteins, are derived from the original trastuzumab antibody. However, the constant regions have been modified to make it more similar to the human immune system, reducing the risk of immune reactions.
Activity of Runimotamab Biosimilar
Runimotamab Biosimilar works by binding to the HER2 receptor and the CD3E protein on the surface of cancer cells. The HER2 receptor is overexpressed in some types of cancer, including breast, gastric, and ovarian cancer. This overexpression leads to uncontrolled cell growth and proliferation. By binding to the HER2 receptor, Runimotamab Biosimilar blocks the signaling pathways that promote cancer growth, inhibiting tumor growth.
In addition, Runimotamab Biosimilar also binds to the CD3E protein, which is found on the surface of T cells. This binding activates the T cells, triggering an immune response against the cancer cells. This dual mechanism of action makes Runimotamab Biosimilar a potent targeted therapy for HER2-positive cancers.
Application of Runimotamab Biosimilar
Runimotamab Biosimilar is currently being used in research grade for the treatment of HER2-positive cancers, particularly breast cancer. It is being studied in clinical trials as a potential alternative to trastuzumab, with the aim of providing a more affordable treatment option.
In addition, Runimotamab Biosimilar is also being investigated for the treatment of other HER2-positive cancers, such as gastric and ovarian cancer. It has shown promising results in preclinical studies and is expected to enter clinical trials for these indications in the near future.
Conclusion
In summary, Runimotamab Biosimilar is a recombinant humanized monoclonal antibody that targets the HER2 receptor and the CD3E protein on cancer cells. Its structure, derived from the original trastuzumab antibody, and its dual mechanism of action make it a potent targeted therapy for HER2-positive cancers. As a biosimilar, it has the potential to provide a more affordable treatment option for patients. Ongoing research and clinical trials are expected to further establish the efficacy and safety of Runimotamab Biosimilar in the treatment of HER2-positive cancers.
There are no reviews yet.