Gemtuzumab Biosimilar: A Promising Antibody for Targeting CD33 in
Cancer Therapy
Gemtuzumab Biosimilar is a monoclonal antibody (mAb) that has been developed as a biosimilar version of the FDA-approved drug Gemtuzumab Ozogamicin (GO). It is a humanized anti-CD33 mAb that specifically targets and binds to the CD33 antigen, which is highly expressed on the surface of cancer cells. Gemtuzumab Biosimilar has shown promising results in preclinical studies and is currently being evaluated in clinical trials as a potential therapeutic option for various types of cancer.
Structure of Gemtuzumab Biosimilar
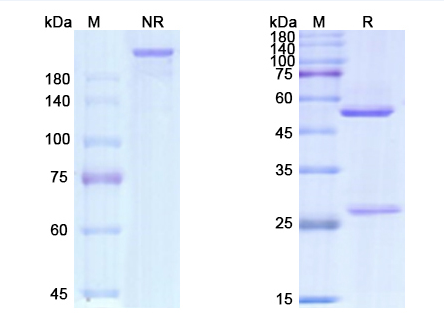
Gemtuzumab Biosimilar is a recombinant humanized IgG4 mAb, with a molecular weight of approximately 148 kDa. It is composed of two identical heavy chains and two identical light chains, connected by disulfide bonds. The heavy chains consist of four constant domains (CH1, CH2, CH3, and CH4) and one variable domain (VH), while the light chains contain one constant domain (CL) and one variable domain (VL). The variable domains of both heavy and light chains are responsible for the antigen-binding specificity of the antibody.
The CD33 binding site of Gemtuzumab Biosimilar is located on the VH and VL domains, which form the antigen-binding fragment (Fab) of the antibody. The constant domains of the heavy chains (CH2 and CH3) are responsible for the effector functions of the antibody, such as antibody-dependent cellular cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC).
Mechanism of Action
Gemtuzumab Biosimilar binds to the CD33 antigen on the surface of cancer cells, leading to the internalization of the antibody-antigen complex via receptor-mediated endocytosis. Once inside the cell, the antibody is cleaved by lysosomal enzymes, releasing the cytotoxic drug calicheamicin. Calicheamicin binds to DNA and causes double-strand breaks, ultimately leading to cell death. This targeted delivery of the cytotoxic drug minimizes its systemic toxicity and improves its efficacy against cancer cells.
In addition to its direct cytotoxic effects, Gemtuzumab Biosimilar also activates the immune system through its Fc region, leading to the destruction of cancer cells by immune cells. This mechanism of action makes Gemtuzumab Biosimilar a potent therapeutic option for cancer treatment.
Applications of Gemtuzumab Biosimilar
Gemtuzumab Biosimilar is being studied for its potential use in the treatment of various types of cancer, including acute myeloid leukemia (AML), myelodysplastic syndrome (MDS), and acute lymphoblastic leukemia (ALL). CD33 is highly expressed on the surface of leukemic cells in these types of cancer, making it an ideal target for Gemtuzumab Biosimilar.
In preclinical studies, Gemtuzumab Biosimilar has shown promising results in inhibiting the growth and survival of cancer cells, both in vitro and in vivo. It has also demonstrated synergistic effects when combined with other chemotherapeutic agents, making it a potential candidate for combination therapies.
Conclusion
Gemtuzumab Biosimilar is a promising antibody that specifically targets the CD33 antigen on cancer cells. Its unique mechanism of action, with both direct cytotoxic effects and immune-mediated effects, makes it a potential therapeutic option for various types of cancer. Further clinical trials are needed to evaluate the safety and efficacy of Gemtuzumab Biosimilar, but it holds great promise in the field of cancer therapy.
Keywords:
Gemtuzumab Biosimilar, anti-CD33 mAb, antibody, therapeutic target, cancer therapy, monoclonal antibody, biosimilar, CD
There are no reviews yet.