Introduction to Vudalimab Biosimilar – Anti-CTLA4,PDCD1 mAb – Research Grade
Vudalimab Biosimilar is a novel antibody that targets two important immune checkpoint proteins, CTLA-4 and PDCD1, which play a crucial role in regulating the immune response. This biosimilar is a promising therapeutic agent that has the potential to treat a wide range of diseases, including cancer and autoimmune disorders.
Structure of Vudalimab Biosimilar
Vudalimab Biosimilar is a monoclonal antibody (mAb) that is produced using recombinant DNA technology. It is a fully humanized antibody, meaning that it is derived from human cells, making it less likely to cause an immune reaction in patients. The antibody has a molecular weight of approximately 150 kDa and consists of two heavy chains and two light chains. The heavy chains are linked to the light chains by disulfide bonds, forming a Y-shaped structure.
Activity of Vudalimab Biosimilar
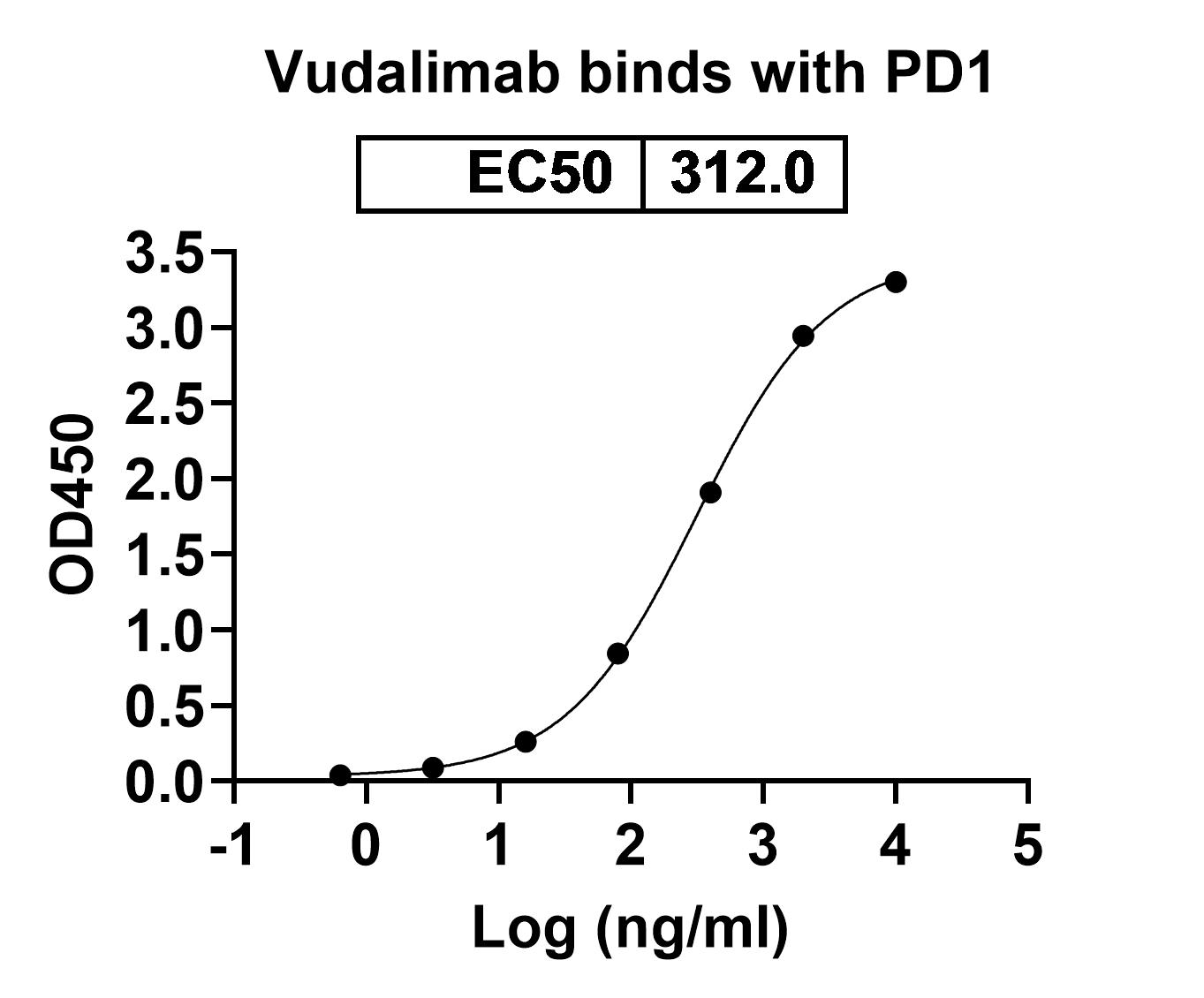
Vudalimab Biosimilar works by binding to and inhibiting two immune checkpoint proteins, CTLA-4 and PDCD1. These proteins are expressed on the surface of T cells, which are a type of white blood cell that plays a key role in the immune response. When activated, CTLA-4 and PDCD1 act as brakes on the immune system, preventing an excessive immune response. However, in diseases such as cancer, these proteins are overexpressed, leading to a weakened immune response. Vudalimab Biosimilar blocks the activity of CTLA-4 and PDCD1, allowing the immune system to mount a stronger response against cancer cells.
Application of Vudalimab Biosimilar
Vudalimab Biosimilar has shown promising results in preclinical studies and is currently being evaluated in clinical trials for the treatment of various types of cancer, including melanoma, lung cancer, and bladder cancer. It is also being investigated as a potential treatment for autoimmune disorders such as rheumatoid arthritis and lupus.
As a research grade product, Vudalimab Biosimilar is also widely used in laboratory research to study the role of CTLA-4 and PDCD1 in various diseases. Its high specificity and potency make it a valuable tool for understanding the immune response and developing new therapies.
Advantages of Vudalimab Biosimilar
Compared to other anti-CTLA-4 and PDCD1 antibodies, Vudalimab Biosimilar has several advantages. As a fully humanized antibody, it has a lower risk of causing an immune response in patients. It also has a longer half-life, meaning it stays in the body for a longer period, allowing for less frequent dosing. Additionally, Vudalimab Biosimilar has a high binding affinity for both CTLA-4 and PDCD1, making it a potent inhibitor of these immune checkpoint proteins.
Future potential of Vudalimab Biosimilar
With the growing understanding of the role of immune checkpoint proteins in various diseases, the potential of Vudalimab Biosimilar as a therapeutic agent is vast. It has the potential to be used in combination with other therapies, such as chemotherapy and radiation, to enhance their effectiveness. Furthermore, ongoing research is exploring the use of Vudalimab Biosimilar in combination with other immune checkpoint inhibitors, which may lead to even more significant clinical benefits.
In conclusion, Vudalimab Biosimilar – Anti-CTLA4,PDCD1 mAb – Research Grade is a promising therapeutic agent that targets two important immune checkpoint proteins, CTLA-4 and PDCD1. Its unique structure and potent activity make it a valuable tool for both research and clinical use. With ongoing studies and trials, Vudalimab Biosimilar has the potential to make a significant impact in the treatment of cancer and autoimmune disorders.
There are no reviews yet.