Cart (0 Items)
Your cart is currently empty.
View ProductsIt looks like you are visiting from outside the EU. Switch to the US version to see local pricing in USD and local shipping.
Switch to US ($)€185.00
100µg 0% OFF + 185 loyalty points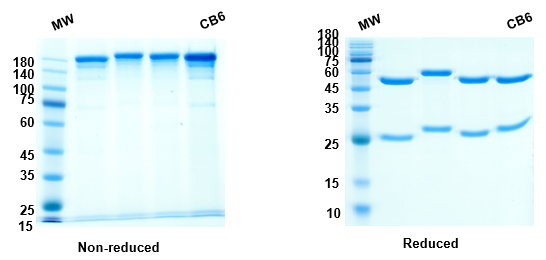

Anti-RBD-1 (Etesevimab) antibody, on SDS-PAGE under reducing and non-reducing conditions. The gel was stained overnight with Coomassie Blue. The purity of the protein is greater than 95%.

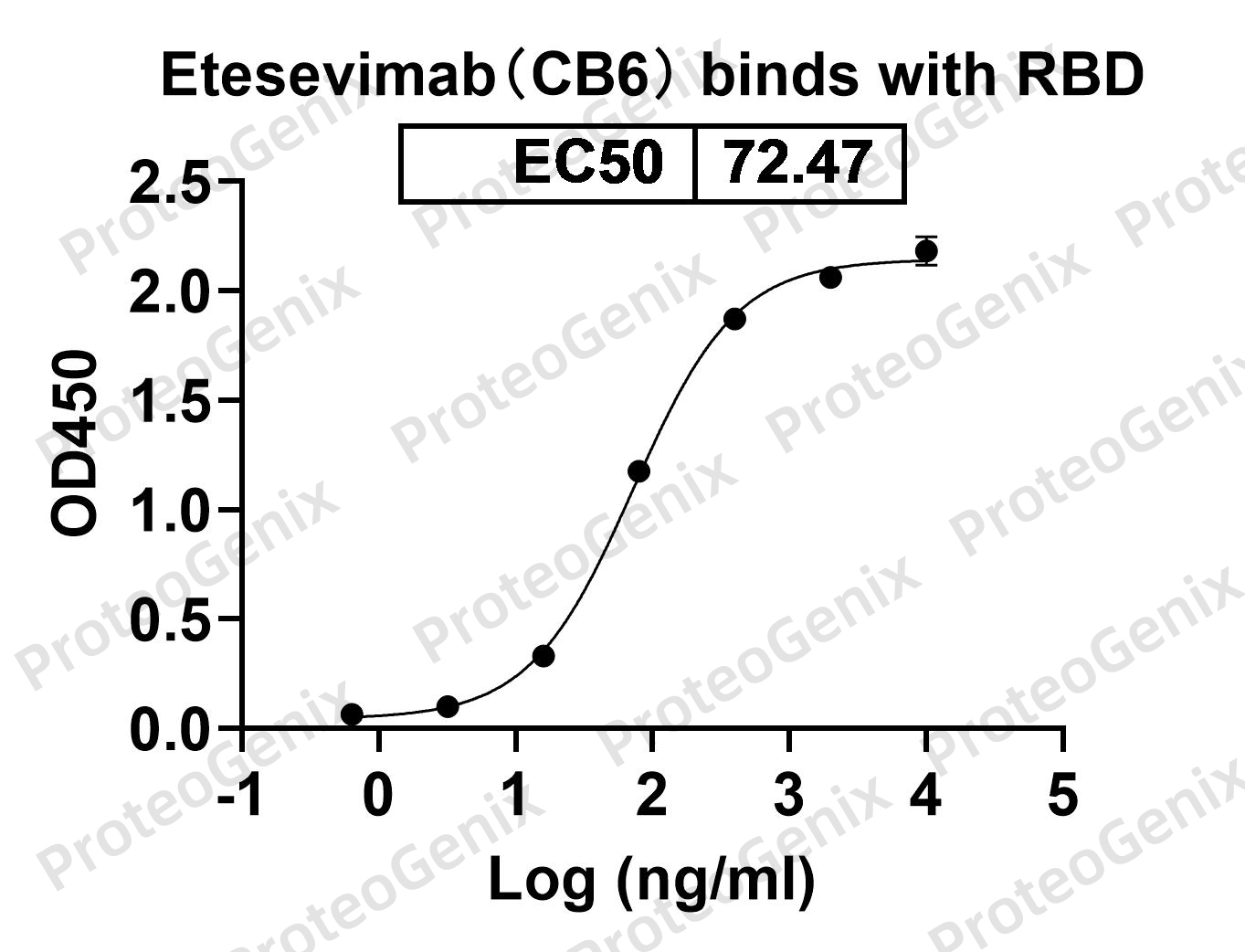
Immobilized RBD Domain (cat. No.PX-COV-P046) at 0.5µg/mL (100µL/well) can bind to Anti-RBD-1 (Etesevimab) antibody (cat. No.PTXCOV-A549) in indirect ELISA with Goat Anti-Human IgG secondary antibody coupled with HRP measured by OD450
Our catalog doesn’t cover everything — but our team does. Whether you need a custom antibody, a specific protein variant, or a bulk order, our scientists are here to help.
There are no reviews yet.