Tilogotamab Biosimilar – Anti-TNFRSF10B mAb – Research Grade
Tilogotamab Biosimilar – Anti-TNFRSF10B mAb – Research Grade: A Promising Antibody for Targeting TNFRSF10B
Introduction
Tilogotamab Biosimilar is a monoclonal antibody (mAb) that specifically targets TNFRSF10B, also known as Death Receptor 5 (DR5). This biosimilar is a highly effective therapeutic agent that has shown promising results in preclinical studies and is currently being evaluated in clinical trials. In this article, we will explore the structure, activity, and potential applications of Tilogotamab Biosimilar as an anti-TNFRSF10B mAb.
Structure of Tilogotamab Biosimilar
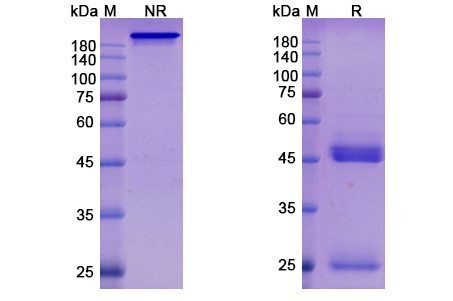
Tilogotamab Biosimilar is a fully human IgG1 mAb that is produced using recombinant DNA technology. It is composed of two heavy chains and two light chains, each containing a variable region and a constant region. The variable region of the antibody is responsible for binding to TNFRSF10B, while the constant region determines the effector functions of the antibody.
Mechanism of Action
TNFRSF10B is a cell surface receptor that belongs to the tumor necrosis factor (TNF) receptor superfamily. It is expressed on the surface of various cancer cells and plays a crucial role in promoting tumor cell survival and proliferation. Tilogotamab Biosimilar binds to TNFRSF10B with high affinity and blocks its interaction with its ligand, TRAIL (TNF-related apoptosis-inducing ligand). This prevents the activation of downstream signaling pathways that promote tumor growth and survival, ultimately leading to tumor cell death.
Application of Tilogotamab Biosimilar
Targeting TNFRSF10B in
Cancer Therapy
Tilogotamab Biosimilar has shown promising results in preclinical studies as a potential anti- cancer therapy. It has been demonstrated to be effective against a wide range of tumor types, including breast, lung, colon, and pancreatic cancer. In addition, it has been shown to have synergistic effects when combined with other anti- cancer agents, making it a promising candidate for combination therapy.
Research Grade Antibody for Studying TNFRSF10B
Tilogotamab Biosimilar is also a valuable tool for studying TNFRSF10B in research settings. Its high specificity and affinity for TNFRSF10B make it a suitable antibody for detecting and quantifying the expression of this receptor in various cancer cell lines and tissues. It can also be used to investigate the role of TNFRSF10B in cancer progression and to identify potential therapeutic targets for drug development.
Clinical Trials
Tilogotamab Biosimilar is currently being evaluated in clinical trials for the treatment of various solid tumors, including breast, lung, and ovarian cancer. The results from these trials have shown promising efficacy and safety profiles, and the biosimilar has been well-tolerated by patients. If successful, Tilogotamab Biosimilar has the potential to become a new treatment option for cancer patients.
Conclusion
Tilogotamab Biosimilar is a promising anti-TNFRSF10B mAb that has shown potential as a therapeutic agent in preclinical studies and is currently being evaluated in clinical trials. Its unique mechanism of action and high specificity make it a valuable tool for targeting TNFRSF10B in cancer therapy and research. With further development and clinical validation, Tilogotamab Biosimilar has the potential to become a valuable addition to the arsenal of anti- cancer agents.
There are no reviews yet.