Introduction
Belantamab Biosimilar is a monoclonal antibody (mAb) that targets the tumor necrosis factor receptor superfamily member 17 (TNFRSF17 or CD269). This protein is also known as B-cell maturation antigen (BCMA) and is expressed on the surface of malignant plasma cells, making it a promising therapeutic target for the treatment of multiple myeloma. Belantamab Biosimilar is a research grade antibody that has shown promising results in preclinical studies and is currently being evaluated in clinical trials.
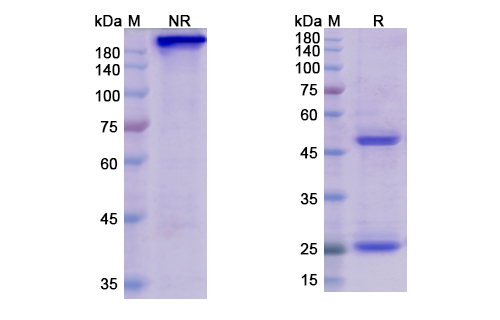
Structure of Belantamab Biosimilar
Belantamab Biosimilar is a fully humanized IgG1 monoclonal antibody. It consists of two heavy chains and two light chains, each with a molecular weight of approximately 50 kDa. The heavy chains contain four constant domains (CH1, CH2, CH3, and CH4) and one variable domain (VH), while the light chains contain two constant domains (CL and CL2) and one variable domain (VL). The variable domains of the heavy and light chains come together to form the antigen-binding site, which specifically recognizes and binds to TNFRSF17.
Mechanism of Action
Belantamab Biosimilar works by binding to TNFRSF17 on the surface of malignant plasma cells, leading to their destruction. This protein is involved in the survival and proliferation of plasma cells, and its overexpression has been linked to the development and progression of multiple myeloma. By targeting TNFRSF17, Belantamab Biosimilar inhibits its signaling pathways, resulting in the induction of cell death and inhibition of tumor growth.
Applications of Belantamab Biosimilar
Belantamab Biosimilar is being developed as a potential treatment for multiple myeloma, a type of cancer that affects plasma cells in the bone marrow. It is intended for use in patients who have relapsed or are refractory to other treatments, such as proteasome inhibitors and immunomodulatory drugs. Belantamab Biosimilar has shown promising results in preclinical studies, including potent anti-tumor activity and minimal toxicity. It is currently being evaluated in clinical trials to assess its safety and efficacy in patients with multiple myeloma.
Advantages of Belantamab Biosimilar
As a fully humanized monoclonal antibody, Belantamab Biosimilar has several advantages over other therapeutic options for multiple myeloma. These include a high specificity for TNFRSF17, low immunogenicity, and a long half-life in the body. Additionally, Belantamab Biosimilar has the potential to be used in combination with other therapies, such as chemotherapy and immunomodulators, to enhance its anti-tumor effects.
Clinical Development of Belantamab Biosimilar
Belantamab Biosimilar is currently in phase I/II clinical trials, with multiple ongoing studies evaluating its safety, efficacy, and dosing in patients with multiple myeloma. The results from these trials will provide valuable information on the potential of Belantamab Biosimilar as a therapeutic option for this disease. If successful, it has the potential to become an important addition to the treatment armamentarium for multiple myeloma.
Conclusion
In summary, Belantamab Biosimilar is a promising research grade antibody that specifically targets TNFRSF17, a protein involved in the development and progression of multiple myeloma. Its unique mechanism of action and potential for use in combination with other therapies make it a promising therapeutic option for patients with relapsed or refractory multiple myeloma. Ongoing clinical trials will provide further insight into the safety and efficacy of Belantamab Biosimilar and its potential to improve outcomes for patients with this disease.
There are no reviews yet.