 Antibody-drug conjugates
Antibody-drug conjugates
ADC payloads
Antibody-drug conjugates (ADCs) are an emerging class of pharmaceuticals designed to deliver highly toxic cargoes (payloads) to clinical targets. In ADCs, payloads are the pharmaceutically active component. For this reason, they dictate the overall effectiveness of these therapies. Up to recently, the development of payloads for ADCs has focused almost exclusively on cytotoxic drugs and cancer targets. But as the safety and efficacy of linker chemistry continue to increase, so does the diversity of payloads that can be used for the generation of new ADCs.
What is a drug payload?
Payloads are one of three primary components of an ADC. They are highly active and toxic pharmaceutical molecules attached to an antibody via a chemical linker. Payloads can be small molecules, protein toxins, biologically active peptides, enzymes, or even radionuclides.
The potency of an antibody-drug conjugate (ADC) is dictated by the final concentration of its cytotoxic payload in tumor cells and tissues. In turn, this concentration depends on the distribution of the payload on the surface of the ADC, conjugate stability, and overall cytotoxicity of the drug. The selection of the proper payload for an ADC depends on the target’s nature, structure, and distribution, among other properties.
How to select a payload for an ADC?
Early ADCs used drug payloads that had been previously approved for clinical use. However, these drugs led to suboptimal results due to their low toxicity, casting research on ADCs in a new direction. Researchers soon discovered that the best payloads for ADCs were the ones that proved to be too toxic for non-targeted clinical applications.
At present, the most efficient payloads are the ones able to kill cancer cells at the nanomolar or picomolar range. Moreover, to be efficient, payloads also need to be fairly soluble, non-immunogenic (small), and possess reactive sites available for conjugation with a linker. Finally, the drug must be compatible with the linker chemistry of the ADC: cleavable or non-cleavable.
Most payloads can be conjugated to cleavable linkers because these molecular bridges are removed in proximity to the target as a response to a chemical or physical signal. In contrast, non-cleavable linkers are stably attached to their payloads. Thus, the presence of the linker must be taken into account when estimating the toxicity of the payload because this type of chemistry often impacts the structure and activity of cytotoxic drugs.
What are the most commonly used cytotoxic payloads in ADC development?
The majority of cytotoxic payloads developed for ADCs belong to two major families: tubulin inhibitors (maytansinoids, auristatins, or taxol derivates) and DNA-modifying agents (mainly calicheamicins). All of these drugs display potent cytotoxicity in the nano and picomolar range – too toxic to be administered systemically.
Tubulin inhibitor payloads act during cell growth by causing cell death via mitotic arrest. They represent the major class of payloads due to their favorable biochemical properties. The most well-known compounds of this class are monomethyl auristatin E (MMAE) and F (MMAF), both synthetic compounds derived from dolastatin 10, a natural antimitotic drug. Presently, several approved ADCs carry MMAE including brentuximab vedotin, polatuzumab vedotin, and enfortumab vedotin.
In contrast, DNA-damaging agents act independently of the cell’s growth process. They have often been described as molecular scissors due to their ability to cleave genomic DNA, causing cell death as a result. Other types of drug payloads currently under active clinical development are RNA-modifying drugs (amatoxins), protein toxins, antibiotics, and enzymes.
| Payload class | Mechanism of action | Examples |
|---|---|---|
| Auristatins | Tubulin polymerase inhibitor | Monomethyl auristatin E (MMAE) and Monomethyl auristatin F (MMAF) |
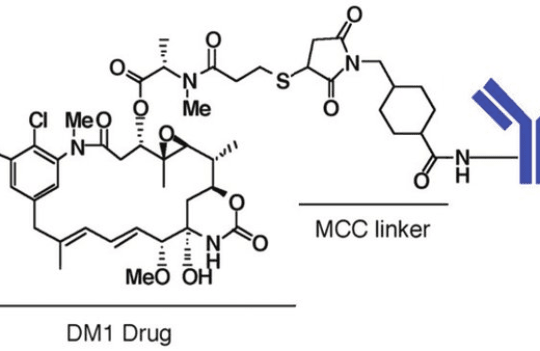
| Maytansines | Tubulin depolymerization | Maytansinoids (DM1 and DM4) |
| Calicheamicins | DNA cleavage | Calicheamicin γ1 |
| Duocarymycins | DNA minor groove alkylating agent | CC-1065 and duocarmycin SA |
| Pyrrolobenzodiazepines (PBD) dimers | DNA minor groove cross-linker | PBD derivates |
| Amatoxins | RNA polymerase II inhibitor | α-Amanitin |
| Protein toxins or Immunotoxins | Several | Pseudomonas exotoxin (PE) and diphtheria toxin (DT) |
| Antibiotics | Several | 4-dimethylamino piperidino-hydroxybenzoxazino rifamycin (dmDNA31) |
| Enzymes | Several | β-glucuronidase, urease, among others |
Generally, the term ADC refers to the conjugation between antibodies and small drugs. The last three classes of payloads are composed by large molecules that pose different challenges due to the hindered pharmacodynamics and diverse mechanisms of action. The conjugation with antibiotics, for instance, is considered an emerging subclass of ADCs and is often named antibody-antibiotic conjugates (AAC). These conjugates arose due to the increasing prevalence of multi-drug resistant bacteria and to the dwindling antibiotic pipeline. AACs provide an alternative to conventional antibiotic therapies due to their specificity, allowing targeting specific groups of pathogenic bacteria without disturbing the healthy microbiota.
Likewise, the conjugation with enzymes is frequently named antibody-enzyme conjugates. These conjugates often employ a two-step strategy comprising the use of a prodrug in tandem with the conjugate. This prodrug acts as a non-toxic precursor that can be administered systemically and only converted to its toxic form in the presence of the enzymatic moiety of the ADC. The development of enzyme conjugates independent of prodrugs is currently underway.
Both enzyme and antibiotic conjugates are considered independent of conventional ADC development due to the unique set of challenges imposed by large, bioactive molecules.
These payloads can be delivered to their specific targets as free drugs (i.e., when using cleavable linker technology), as linker-payload complexes (i.e., when using non-cleavable linker technology), or encapsulated within a nanocarrier. The use of nanocarriers in ADC development is a relatively new area of research. These carriers include lipid-based particles such as liposomes and polymeric molecules. They are designed to encapsulate a high number of drugs per ADC and enhance their pharmacokinetics, distribution, protect drugs from premature degradation, increase the solubility of hydrophobic drugs, and enhance their diffusion across the cell membrane.
In sum, nanocarriers seek to mitigate the problem imposed by the limited number of conjugation sites available on the surface of a monoclonal antibody, while maximizing the concentration each antibody can deliver to its intended target. However, for the moment, antibody-nanocarrier conjugates have yet to reach the market.
The future of ADC payloads
One of the greatest challenges in ADC development is balancing payload efficacy with ADC safety. These two properties depend not only on the inherent toxicity of the payload but also on the drug-to-antibody ratio (DAR) and stability of the linker chemistry. Several approaches are currently under development in an attempt to increase the synergy between antibodies and small drug payloads. Most of these strategies converge around the improvement of linker chemistry and on the combination of ADC with other therapies such as immune checkpoint inhibitors.
However, the development of novel payloads has lagged in comparison to these other fields of research. The market is heavily dominated by auristatins and maytansinoids derivates with PBD dimers slowly gaining ground in ADC applications. The reason for the restricted choice of payloads for ADC development is tied to constraints in terms of synthesis. The availability of precursors for the rapid synthesis of payloads is still the most limiting step of this process.
For the moment, experts believe that improvements in terms of payloads should focus on the development of cost-effective precursors for rapid synthesis and enhanced drug solubilization mechanisms (e.g., use of nanocarriers).
- Criscitiello, C. et al. Antibody-drug conjugates in solid tumors: a look into novel targets. J Hematol Oncol. 2021; 14(1):20. doi: 10.1186/s13045-021-01035-z
- Tang, H. et al. The Analysis of Key Factors Related to ADCs Structural Design. Front Pharmacol. 2019; 10: 373. doi: 10.3389/fphar.2019.00373
You could also be interested in: