Antibody-drug conjugates
Antibody-drug conjugates
New and promising trends in therapeutic ADC generation
With the growth of the global market for therapeutic antibody-drug conjugates (ADCs), researchers are set on developing exciting new technologies. In the next decades, we will likely witness significant changes in the ADC production process. These changes will be accompanied by a better understanding of cancer biology and the identification of new disease markers which, in turn, will allow the production of highly efficient, specific, and safe ADC therapeutics.
What are the future development opportunities for ADCs?
The global market for antibody-drug conjugates (ADCs) is anticipated to reach $7.5 billion by 2025. Kadcyla alone, an anti-HER2 ADC, surpassed $1 billion in annual sales in 2019, becoming the first ADC to hit blockbuster status. Historically, ADCs have been more successful in the treatment of hematological conditions. However, the increase in sales has prompted the diversification of targets, production processes, and mechanisms of action.
These emerging biotherapeutics conjugate the specificity of immunotherapies with the potency of chemotherapeutics, allowing the delivery of extremely toxic drugs to cancer cells with high precision. The chemistry behind the production of ADCs is complex and undergoing active development. Currently, major trends in the clinical pipeline hint at a shift in the preferred structures, chemistries, and cargoes of ADCs, intended to make the therapy safer and broaden its therapeutic window.
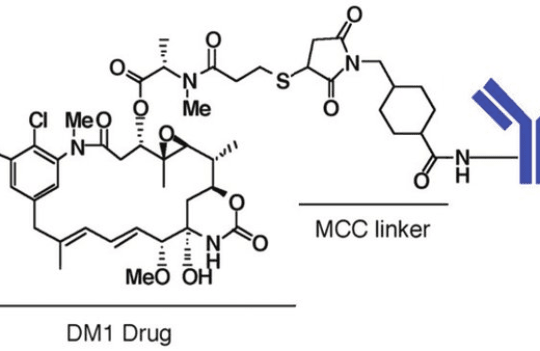
ADCs consist of an antibody carrier (monoclonal, bispecific, or antibody fragment) conjugated to a payload (small drug, enzyme, antibiotic, etc.) via a cleavable or non-cleavable linker. Early ADCs suffered from considerable problems in terms of safety caused by off-target payload release or heterogeneity due to poorly efficient conjugations chemistry.
In an attempt to solve these issues, several strategies are currently under active development:
- Site-specific and click chemistry are quickly replacing heterogeneity-prone methods such as cysteine and lysine-based conjugation.
- New conjugation chemistry is also allowing the safe increase of DAR (drug-to-antibody ratio) values, consequently permitting the use of less cytotoxic agents and thus widening the therapeutic window.
- The improvement of cleavable linker chemistry continues to enhance the bystander effect (killing of antigen-negative cells in the tumor microenvironment) allowing the better treatment of solid tumors and making ADC therapeutics more resistant to mutations in tumor-associated receptors.
- The development of probody drug conjugates has the potential to significantly increase the safety of these therapeutics. In this case, the drug circulates in an inactivated form and it is only activated in the tumor microenvironment by specific proteases.
- Bispecific and biparatopic antibodies targeting non-overlapping epitopes or paratopes are also under current development as carriers (bsADCs). Studies show that targeting a single receptor often leads to drug resistance. Moreover, targeting two different markers of the same disease pathway has shown to have a synergistic effect.
- With the purpose of fighting drug resistance and handling target heterogeneity, dual-drug ADCs are also under active development.
Challenges and opportunities in anti-cancer ADC therapies
Three of the most important challenges faced by ADC therapeutics are tied to the treatment of solid tumors, tumor-initiating cells (TICs), and senescent cells.
Solid tumors are notoriously hard to eradicate given the limited penetration of ADC therapies. For this reason, active fields of research continue to explore new mechanisms of action including the use of smaller antibody carriers that have better tissue diffusion rates (VHH or monoclonal antibody fragments) or employment of more efficient cleavable linkers able to release the toxic cargo in the tumor’s microenvironment as quickly as possible, thus enhancing the bystander effect.
In contrast, TICs are hard to target given the low abundance and high heterogeneity of disease markers on their surface. These cells can be divided in two subpopulations: proliferating and quiescent. For this reason, drugs that act independently of the cell cycle such as DNA or RNA damaging agents, have an advantage over the growth-dependent tubulin inhibitors for targeting and eliminating these pervasive cells. Moreover, given the low abundance of specific TIC markers, the use of highly potent cytotoxic agents with lower DAR values should be preferred. Up to now, conventional chemotherapy, immunotherapy, and ADCs have found limited success in the inhibition of TICs in animal models. Experts point out the lack of suitable models and a poor understanding of TIC biology as the main reasons for failure. Future studies need to focus on casting new light into TIC biology and screening for new specific markers before more progress can be made on the development of new therapies.
Senescent or non-proliferating cells have been linked to multiple conditions ranging from fibrosis, diabetes, and cancer, among others. Senescence is an irreversible mechanism consisting in the arrest of the proliferation process, leading to the accumulation of these cells. In turn, their progressive accumulation results in the impairment of tissue functions and plays an important role in tumor progression. Historically, it has been extremely hard to target these cells due to their non-proliferating stage. However, that may soon change due to the recent description of the surfaceome (surface cell receptors) of senescent cells and the development of ADCs able to recognize them with great precision.
Concluding remarks
ADC development and production are active fields of research. With the rise of alternative chemistries, conjugation technologies, and payloads, ADCs are on the track to tackle increasingly complex diseases and challenges. Experts believe that the demand for these therapeutics will continue to rise and that treatment of solid tumors and non-proliferating cells in the clinical setting might soon reach better efficiencies.
- Criscitiello, C. et al. Antibody–drug conjugates in solid tumors: a look into novel targets. J Hematol Oncol. 2021; 14(1):20. doi: 10.1186/s13045-021-01035-z
- Marcucci, F. et al. Antibody-Drug Conjugates (ADC) Against Cancer Stem-Like Cells (CSC)—Is There Still Room for Optimism? Front Oncol. 2019; 9: 167. doi: 10.3389/fonc.2019.00167
- Poblocka, M. et al. Targeted clearance of senescent cells using an antibody-drug conjugate against a specific membrane marker. Sci Rep. 2020; 11:20358. doi: 10.1038/s41598-021-99852-2
- Shim, H. Bispecific Antibodies and Antibody–Drug Conjugates for Cancer Therapy: Technological Considerations. Biomolecules. 2020; 10(3): 360. doi: 10.3390/biom10030360
- Thirlway, J. ADCs and solid tumors – The payload revolution that drives therapeutic opportunities in unmet need. Drug Discovery World. 2020; 21(2). Retrieved from: https://www.ddw-online.com/adcs-and-solid-tumours-the-payload-revolution-that-drives-therapeutic-opportunities-in-unmet-need-848-202004/
- Yamazaki, C. M. et al. Antibody-drug conjugates with dual payloads for combating breast tumor heterogeneity and drug resistance. Nat Commun. 2021; 12(1):3528. doi: 10.1038/s41467-021-23793-7
You could also be interested in: