 Antibody-drug conjugates
Antibody-drug conjugates
What are the most common targets for ADCs?
Currently, dozens of antibody-drug conjugates (ADCs) and immunotoxins have reached the clinic or are under active clinical development. These invaluable ADC therapies target a plethora of disease markers. In this article, we provide an overview of the most common targets and analyze their underlying key properties.
Common targets of therapeutic ADCs
Antibody-drug conjugates (ADCs) are invaluable biotherapeutics successfully used to treat several forms of cancer. These biomolecules combine the specificity of antibodies with the cytotoxic potency of small drugs, creating an efficient and safe delivery system for molecules that are too toxic for systemic administration. Currently, 12 therapeutic ADCs received marketing approval from the FDA. They target a variety of disease markers that can be found in the table below.
| Drug | Trade name | Target | Format | Condition | FDA approval |
|---|---|---|---|---|---|
| Tisotumab vedotin | Tivdak | Tissue factor (TF) | Human IgG1 ADC | Cervical cancer | 2021 |
| Loncastuximab tesirine | Zynlonta | CD19 | Humanized IgG1 ADC | Diffuse large B-cell lymphoma | 2021 |
| Belantamab mafodotin | Blenrep | BCMA | Humanized IgG1 ADC | Multiple myeloma | 2020 |
| Sacituzumab govitecan | Trodelvy | TROP-2 | Humanized IgG1 ADC | Triple-negative breast cancer | 2020 |
| Trastuzumab deruxtecan | Enhertu | HER2 | Humanized IgG1 ADC | HER2+ breast cancer | 2019 |
| Enfortumab vedotin | Padcev | Nectin-4 | Human IgG1 ADC | Urothelial cancer | 2019 |
| Polatuzumab vedotin | Polivy | CD79b | Humanized IgG1 ADC | Diffuse large B-cell lymphoma | 2019 |
| Moxetumomab pasudotox | Lumoxiti | CD22 | Murine IgG1 dsFv immunotoxin | Hairy cell leukemia | 2018 |
| Inotuzumab ozogamicin | Besponsa | CD22 | Humanized IgG4 ADC | Hematological malignancy | 2017 |
| Trastuzumab emtansine | Kadcyla | HER2 | Humanized IgG1 ADC | HER2+ breast cancer | 2013 |
| Brentuximab vedotin | Adcetris | CD30 | Chimeric IgG1 | Hodgkin lymphoma, systemic anaplastic large cell lymphoma | 2011 |
| Gemtuzumab ozogamicin | Mylotarg | CD33 | Humanized IgG4 ADC | Acute myeloid leukemia | 2017; 2000 |
HER2 as a major target of anti-cancer ADCs
An analysis of ADCs currently in the clinical pipeline revealed that HER2 remains one of the most targeted cancer markers. HER2 stands for receptor tyrosine-protein kinase erbB-2, also called CD340 (cluster of differentiation 340), proto-oncogene Neu, or ERBB2 protein. It belongs to the family of human epidermal growth factor receptors (HER/EGFR/ERBB) and its overexpression is known to correlate with the progression of specific types of aggressive breast cancers.
It is found in approximately 30% of breast cancer patients, making it an invaluable biomarker for both treatment and diagnostics. Historically, HER2-positive cancer has been associated with poor prognosis with a median overall survival of 15 months when employing conventional chemotherapy in the metastatic phase. Although conventional anti-HER2 immunotherapies have proved successful in delaying the progression of the disease, maximal antitumoral activity was found in patients who received a combination of immunotherapy and chemotherapy.
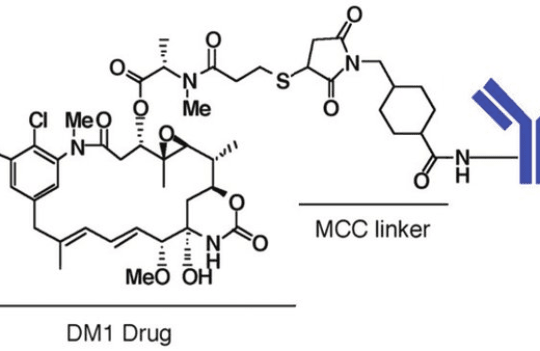
Trastuzumab emtansine (Kadcyla) and Trastuzumab deruxtecan (Enhertu) are the two FDA-approved anti-HER2 ADCs. Despite sharing the same target and antibody backbone, their mechanisms of action could not be more different. For instance, Kadcyla contains a non-cleavable linker attached to a potent microtubule inhibitor, mertansine (DM1) with a drug-to-antibody ratio (DAR) of 3.5. In contrast, Enhertu carries an exatecan derivate (DXd), a potent topoisomerase I inhibitor, conjugated to the antibody via a protease-cleavable linker with a DAR of 8.
Although both therapies have found great success in delaying the progression of the disease, Enhertu provides some advantages including:
- Cleavable linker – allows the release of the payload in the tumor microenvironment, thus, making the treatment more resistant to mutations in the HER2 due to the bystander effect (killing of HER2-negative in the vicinity of HER2-positive cells).
- Growth stage independent – microtubule inhibitors act on actively dividing cells, in contrast, DNA-damaging agents such as DXd are independent of the growth stage of the cell. This is an advantage when targeting dormant cancer that remains recalcitrant to most drugs leading to life-threatening metastasis.
CD22 as a major target of anti-cancer ADCs
There are currently one ADC and one immunotoxin in the clinic designed to target CD22 (cluster of differentiation 22): Moxetumomab pasudotox (Lumoxiti) and Inotuzumab ozogamicin (Besponsa). CD22 is a transmembrane glycoprotein found on the surface of mature B cells. It is responsible for modulating B-cell function, survival, and apoptosis. Most B cell malignancies are known to correlate with the overexpression of CD22, thus, this receptor has become a major therapeutic target for several B-cell diseases including non-Hodgkin lymphoma (NHL), hairy cell leukemia (HCL), Burkitt’s lymphoma, and acute lymphoblastic leukemia (ALL).
Besponsa combines an anti-CD22 monoclonal antibody with calicheamicin, a DNA-damaging agent that causes breaks in the DNA leading to cell death by apoptosis. The two components are conjugated via a cleavable AcBut acyl hydrazone-disulfide acid-cleavable linker. This ADC therapeutic has shown outstanding response in treatment-refractory patients with ALL. In animal studies, the combined drug also proved to be more effective than conventional chemotherapy at inhibiting tumor growth.
In contrast, Lumoxiti is an immunotoxin that combines the Fv portion of an antibody with a fragment of a potent bacterial toxin – Pseudomonas exotoxin A, fragment PE38. This toxin induces cell death by inhibiting protein synthesis. Partial remission and significant tumor inhibition have been described for patients with HCL. However, unlike conventional ADCs, immunotoxins are generally more immunogenic and prone to causing anti-toxin antibody responses, which has been shown to limit treatment efficiency.
Experts believe that there is much room for improvement in anti-CD22 ADC treatments, particularly with the use of safer linkers and more efficient linkers. For this reason, anti-CD22 linker chemistry will evolve from the acid-labile linkers (hydrazone-based), which are prone to off-target cleavage, towards protease-cleavable linkers, which have proved to be more specific and better suited for safe cytotoxic drug delivery.
What makes a good target for ADC biotherapeutics?
Despite the diversity of targets of therapeutic ADCs, these share several key properties:
- Membrane-bound receptors with an extracellular domain
- Limited secretion (absent from circulation)
- Highly expressed in cancer cells, but may be expressed at low levels in quickly regenerating tissues
- Homogeneous expression
- Highly efficient internalization
- Able to undergo the appropriate trafficking route
- Quickly recycled back to the surface by cancer cells
- Chang, L. S. et al. Targeting CD22 in B-cell malignancies: current status and clinical outlook. BioDrugs. 2013; 27(4):293-304. doi: 10.1007/s40259-013-0016-7
- Ferraro, E. et al. Implementing antibody-drug conjugates (ADCs) in HER2-positive breast cancer: state of the art and future directions. Breast Cancer Res. 2021; 23(1):84. doi: 10.1186/s13058-021-01459-y
- Nejadmoghaddam, M. R. et al. Antibody-Drug Conjugates: Possibilities and Challenges. Avicenna J Med Biotechnol. 2019; 11(1): 3–23. Retrieved from ncbi.nlm.nih.gov/pmc/articles/PMC6359697
You could also be interested in: