Antibody-drug conjugates
Antibody-drug conjugates
Bioanalysis of antibody-drug conjugates (ADCs)
The bioanalysis of antibody-drug conjugates remains challenging. However, in recent years, many methodologies have been developed to tackle the unique issues imposed by these heterogeneous therapeutics. In this article, we provide an overview of the key properties and methods currently in use for the extensive and quick characterization of antibody-drug conjugates.
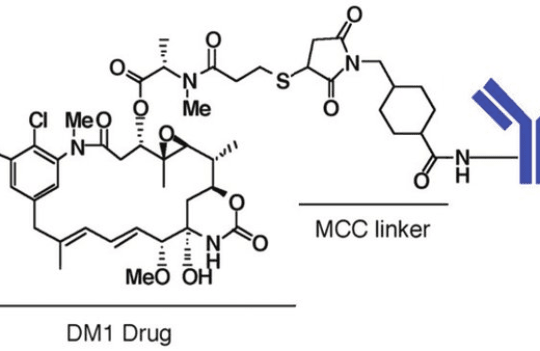
Antibody-drug conjugates (ADCs) consist of small cytotoxic agents attached to an antibody carrier (monoclonal, bispecific, or antibody fragment) via chemical linkers. ADCs are an emerging class of biotherapeutics able to combine the target specificity of antibodies with the potency of chemotherapeutics, and by doing so, they minimize systemic toxicity while enhancing therapeutic potency.
One of the major challenges associated with the production of ADCs lies in their heterogeneous nature. Due to the significant variability introduced by linker chemistry and conjugation processes, ADCs are typically a mixture of different ADC species with a variable number of payloads attached to the surface of the antibody. For this reason, bioanalysis of these conjugates is of extreme importance, not only to ensure reasonable batch-to-batch consistency but also to enhance safety and therapeutic effectiveness.
Key properties of ADCs
Linker chemistry (cleavable and non-cleavable) and conjugation methods (cysteine, lysine, or site-specific conjugation) dictate the efficacy and influence the key properties of ADCs. Cleavable linkers are particularly prone to off-target degradation. For this reason, considerable efforts have been employed into making these linkers more resistant and into shielding sensitive residues from environmental signals.
Moreover, most of the ADCs currently in the clinic or in active clinical development are based on cysteine and lysine conjugation methods. The functionalization of these residues often results in great variability in the number and distribution of drug molecules on the surface of the carrier. Experts agree that a shift towards site-specific conjugation methods might greatly narrow the diversity of ADC species in a single batch.
Currently, the major properties of ADCs and common bioanalytical methods used for their study include:
- Drug-to-antibody ratio (DAR): ultraviolet-visible (UV/Vis) spectroscopy, cathepsin-B enzyme-based digestion method, hydrophobic interaction chromatography (HIC), reversed-phase high-performance liquid chromatography (RP-HPLC), and mass spectrometry (MS)
- Drug load distribution: capillary electrophoresis (CE), HIC, RP-HPLC, and MS
- Unconjugated antibody: CE, ELISA HIC, RP-HPLC, and MS
- Degradation/biotransformation: LC-MS/MS (can be preceded by enrichment and proteolytic degradation steps to increase analytical resolution
- Free drug content: LC-MS/MS
- Charge variants: ion-exchange chromatography (IEX) and imaged capillary isoelectric focusing (iCIEF)
- Stability and aggregation profile: size-exclusion chromatography (SEC), LC-MS, and CE
- Post-translational modifications: LC-MS/MS
DAR and drug load distribution are the two major biochemical properties of ADCs and they will be covered in the section below.
Drug-to-antibody ratio (DAR) and drug load distribution
DAR is defined as the average number of drug molecules conjugated to an antibody in a particular ADC batch. In contrast, drug load distribution is defined as the stoichiometric distribution or range of drug molecules conjugated to an antibody carrier. These properties dictate the therapeutic efficacy of ADCs and their pharmacokinetic profile. For instance, studies show that high DAR values hinder the pharmacokinetics of ADCs and potentially increase the toxicity of ADCs, while low DAR values may result in ADCs with limited therapeutic efficacy. Ideally, DAR values should be between 2 and 4.
Of all the methods mentioned above, UV/Vis, HIC, and RP-HPLC, or LC-MS remain the most widely used for the analysis of DAR values and drug load distribution. However, methods that rely on the principle of capillary electrophoresis continue to gain ground over more conventional methods due to the simple instrumentation, miniaturized format, rapid separation, low sample input, and high resolution.
Bioanalysis of the pharmacokinetics of ADCs
Pharmacokinetics is the study of the fate of a drug in an organism, while pharmacodynamics can be described as the organism’s reaction to the drug. These properties are interdependent and interconnected because the stability of the drug in circulation affects its effective concentration in tumor sites and, subsequently, its therapeutic efficacy. PKPD studies are thus carried out in tandem using animal models of a specific disease to test different dosages and dose regimens and measure physiological responses (PD – tumor reduction and remission, among others) and biotransformation (PK).
Several properties can help estimate the PK profile of ADCs by administrating them to animal models and subsequently collecting their plasma. These properties include:
- Total and conjugated antibodies: typically estimated via modified ELISA where the rate of in vivo deconjugation can be measured by capturing the total antibody followed by the capture of intact ADC complexes.
- Free and conjugated drugs: LC-MS/MS methods are the most sensitive for this type of analysis. This quantification is achieved by a series of pre-treatments that involve protein precipitation and/or cleavage of linkers in vitro.
- Biotransformation: proteolytic degradation of ADCs may render metabolites with cytotoxic activity. For this reason, the careful analysis and structural characterization of these metabolites remain extremely important. Currently, this is achieved via complex multi-step platforms that involve immuno-capture, proteolytic digestion, chromatographic separation, and mass spectrometry.
Monitoring the behavior of ADCs and correlating it with their therapeutic efficacy is vital to ensure the safety of ADC therapies. Studying this data helps to estimate how the ADC therapeutic is likely to behave in clinical studies, ensuring a smooth progression through the clinical pipeline.
Concluding remarks
The biochemical and biophysical properties of ADCs can help predict the therapeutic efficacy and safety of these complex biopharmaceuticals in the clinic. One of the main challenges of bioanalysis lies in the fact that ADCs represent a complex mixture of ADC species with different charges, drug loads, and distribution. This hinders the identification of potentially problematic metabolites and makes the analysis of degradation pathways significantly more complex.
However, technical progress has significantly increased the resolution of certain techniques, making the analysis of complex mixtures possible. Interestingly, current trends in ADC development point out to the adoption of site-specific conjugation techniques (known to reduce drug load variability) and more hydrophilic linkers (known to reduce aggregation). Techniques such as capillary electrophoresis and advanced platforms of bioanalysis are also being established to ensure the quick and extensive characterization of these life-saving biotherapeutics.
- Gorovits, B. Bioanalysis of antibody-drug conjugates. Bioanalysis. 2015; 7(13):1559-15560. doi: 10.4155/bio.15.106
- Mou, S. et al. ADME Considerations and Bioanalytical Strategies for Pharmacokinetic Assessments of Antibody-Drug Conjugates. Antibodies (Basel). 2018; 7(4): 41. doi: 10.3390/antib7040041