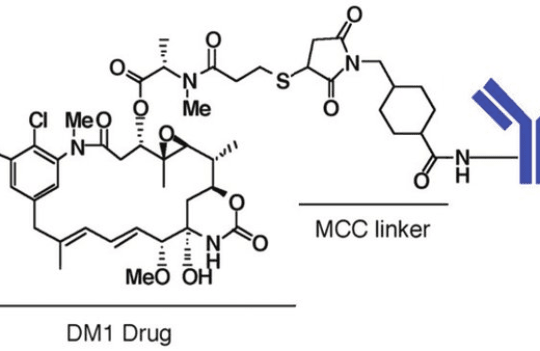
Antibody-drug conjugates
Antibody-drug conjugates
Key biochemical and biophysical properties of antibody-drug conjugates (ADCs)
Antibody-drug conjugates (ADCs) are complex molecules. Their key properties are heavily influenced by linker chemistry and antibody conjugation methods. For this reason, several ADC bioanalysis methods have been developed over the years to guarantee the stability and clinical effectiveness of these biotherapeutics. In this article, we discuss the key biochemical and biophysical properties of ADCs, as well as provide an overview regarding the most widely used analytical methods.
Key biochemical and biophysical properties of ADCs
Antibody-drug conjugates (ADCs) are complex biomolecules with outstanding therapeutic potential. These constructs consist of an antibody carrier (i.e., monoclonal antibody or antibody fragment) and several drug molecules attached to its surface via cleavable or non-cleavable linkers. The efficacy of these biotherapeutics is heavily influenced by linker and conjugation chemistries as well as by, warranting an extensive characterization of several biochemical and biophysical properties of the ADC molecule including: drug load distribution, drug-to-antibody ratio (DAR), unconjugated antibody, charge variants, stability, aggregation, degradation, hydrophobicity profile, and post-translational modifications, among others.
Why is linker chemistry so significant for ADC efficacy? Linkers can incorporate chemical triggers resulting in payload release in specific conditions or the presence of specific enzymes. Cleavable linkers represent the major class of linkers used in ADC development. The most commonly used include peptide linkers (cleaved by cathepsin B) and hydrazone-disulfide-containing linkers (cleaved at low pH values and in reducing conditions). These linkers are often prone to off-site degradation leading to premature release. In contrast, non-cleavable linkers are more stable because they are only degraded upon the internalization of the ADC molecule. However, they often suffer from low therapeutic efficacy due to inefficient payload release, poor cellular trafficking, and modification of the payload’s bioactivity.
DAR, drug load distribution, and unconjugated antibody
These are considered the three key properties of ADCs. DAR is the average drug-to-antibody ratio in a batch of ADCs. It is considered a rough measure and routinely used as a quality control tool to monitor batch-to-batch variability in ADC production processes. In contrast, the term drug load distribution is used to describe a much more precise property. It entails the stoichiometric distribution of drug molecules on the antibody carriers. In other words, DAR is an average measure while drug load distribution is a range of DAR values observed in a single batch. Unconjugated antibody levels are defined as the amount of antibody molecules devoid of conjugated drugs.
Due to the importance of these properties, several methods are recurrently used during ADC development and quality control purposes. These methods range from capillary electrophoresis (CE), hydrophobic interaction chromatography (HIC), and mass spectrometry (MS). All these methods have been covered in great detail elsewhere.
Determination of charge variants of ADCs
Charge variants affect the stability, binding affinity, and, consequently, biological activity of ADCs. These biotherapeutics are heterogeneous by nature and various charge variants are known to co-exist in the same batch caused by linker chemistry, conjugation, post-translational modifications, protein degradation, and even due to the purification process.
Two widely efficient and sensitive methods routinely used to detect subtle charge differences in ADC variants are ion-exchange chromatography (IEX) and imaged capillary isoelectric focusing (iCIEF). Nevertheless, the analysis remains challenging due to the complex nature of ADC molecules. This is especially true for lysine-conjugated ADCs, where lysine residues interfere with the parent antibody charge variant distribution. In contrast, both IEX and iCIEF methods are more useful when analyzing cysteine-conjugated ADCs.
ADC degradation and stability characterization
The stability profile and degradation pathways of ADCs are significantly more complex than those of monoclonal antibodies. Not only do the common antibody degradation mechanisms apply, but also the process of antibody conjugation is known to change the structure, binding properties, and hydrophobicity of the resulting biomolecules. Moreover, some conjugation methods (i.e., lysine-based) often introduce the risk of cross-linkage between different ADC species.
One of the most important properties to measure when determining ADC stability is aggregation propensity. As a general rule, higher DAR increase ADC hydrophobicity and, as a consequence, the risk of aggregation. But, other factors, such as the use of specific reagents such as dimethylformamide (DMF) can also contribute to this phenomenon, widely described for the conjugation process of Mylotarg™. In this case, the aggregation is caused by the use of high concentrations of DMF, and its negative effect was found to be mitigated by using additives such as glycerol, propylene glycol, and octanoic acid. In turn, these additives also allow the use of lower DMF concentrations during conjugation. Another strategy to mitigate the increased hydrophobicity of ADCs is to use and develop more hydrophilic linkers or attach polyethylene glycol (PEG) chains.
Many methods are currently used for physical and chemical stability analysis:
- Size-exclusion chromatography (SEC)
- Liquid chromatography and mass spectrometry (LC-MS)
- Two-dimensional liquid chromatography (2D-LC) – typically a combination of two separation methods such as SEC and RP-HPLC (reverse-phase high-performance liquid chromatography)
- Capillary electrophoresis (CE)
Analysis of post-translational modifications of ADCs
Post Translational Modifications (PTM) such as glycosylation, acylation, and disulfide bridge configuration have a significant impact on the immunogenicity and efficacy of antibodies and corresponding ADC molecules. In fact, the conjugation of payloads with an antibody is considered a type of PTM. For this reason, many methods used to measure PTM in monoclonal antibodies have been successfully used to resolve ADC molecules.
Typically, proteolytic digestion followed by LC or GC-MS (gas chromatography) based methods are routinely used during ADC development to confirm the oligosaccharide profile. This routine analysis performed before and after conjugation helps measure the impact of the linker chemistry on the glycosylation profile of the antibody carrier.
- Wagh, A. et al. Challenges and new frontiers in analytical characterization of antibody-drug conjugates. MAbs. 2018; 10(2):222-243. doi: 10.1080/19420862.2017.1412025