Antibody-drug conjugates
Antibody-drug conjugates
How to choose the best linker for an ADC? Cleavable vs. non-cleavable
Antibody-drug conjugate linker chemistry has greatly evolved since the development of the first molecules. Linkers can be defined as the interface between chemistry and biology. For this reason, they dictate how and when a payload gets released from its antibody carrier. Currently, there are two main types of linker chemistry: cleavable and non-cleavable. Each with specific advantages and limitations making them suitable for distinct applications.
ADC linker technology
In an ADC molecule, linkers are in the interface of chemistry and biology. Consisting of antibody-binding and payload-binding domains, linkers are ultimately responsible for the safety of the ADC, its therapeutic efficiency, mechanism of action, and therapeutic window.
Linker chemistry determines the stability of the ADC and the payload release rate. For this reason, achieving a balance between extracellular stability and payload cleavage efficiency (intra- or extracellular) are the most important factors that should be taken into account when developing a new linker. As shown in an increasing number of studies, the optimal properties of an ADC linker are unique to each cancer and there is no one-size-fits-all solution.
There are currently two main types of linker chemistry: cleavable and non-cleavable. Non-cleavable linkers are characterized by higher stability in circulation and lower off-target activity due to their reduced “bystander effect” – killing of antigen-negative cells. In contrast, cleavable linkers are prized for their versatility and potential to exploit distinct mechanisms of action. Due to their higher “bystander effect”, this type of chemistry is often associated with higher toxicity. However, several studies have shown that the “bystander effect” may be beneficial against certain cancer types, indicating that opting for one chemistry over another should be assessed on a case-by-case basis.
Cleavable linkers
Disulfides and dipeptides are the dominant motifs found in cleavable linkers. These linkers are extremely versatile because they allow the controlled release of the payload intracellularly or in the tumors’ microenvironment. Over the years, several methods have been developed to create linkers with inbuilt triggers allowing its catalysis in the presence of specific signals or enzymes.
Within this vast class of linkers, chemically labile and enzymatic labile linkers are two of the most well-established approaches.
Chemically labile linkers
Acid-labile linkers cleave in response to low pH values (about 4.5) characteristic of lysosomes and endosomes. Despite their usefulness, they are rarely used today due to being limited to payloads with moderate toxicity. In contrast, disulfide linkers are one of the most popular types of cleavable linkers. This type of chemistry makes the linker susceptible to nucleophilic attacks from thiols – as the ones present in glutathione (GSH), a compound that is highly enriched in the cytosol of cancer cells or on tumors microenvironment due to the increased oxidative stress.
Enzyme labile linkers
Valine-citrulline dipeptide linkers are widely used when enzyme-mediated cleavable is envisaged as the preferred release mechanism. This dipeptide can be cleaved by lysosomal enzymes such as cathepsin B that are also known to spill to the extracellular environment in necrotic areas.
Non-cleavable linkers
Non-cleavable linkers offer the possibility to explore a completely different mechanism of action. These linkers are independent of chemical triggers and directly integrate the payload. For this reason, payload release only occurs upon ADC internalization and lysosomal processing. Once the antibody carrier is digested by lysosomal enzymes, the linker-payload pair is released into the intracellular environment and free to exert its cytotoxic activity.
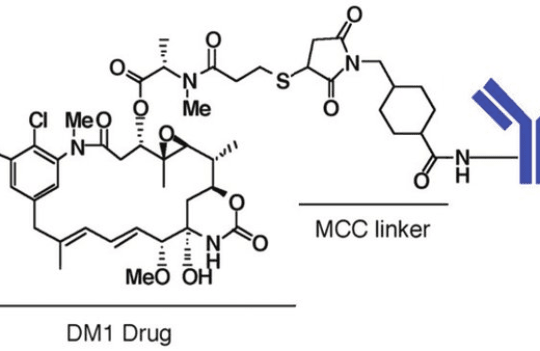
The most important advantage of this approach is the enhanced safety of non-cleavable ADCs. Because they only release their toxic cargoes upon digestion, the risk of off-target toxicity and “bystander effect” are thus minimized. However, this strategy is known to alter the structure and activity of the payload. Most non-cleavable linkers are based on maleimidocaproyl (MC) and 4-maleimidomethyl cyclohexane-1-carboxylate (MCC). More recently, researchers have been developing hydrophilic non-cleavable linkers containing PEG with alkynes and piperazine. However, this novel technology has yet to reach clinical maturity.
Cleavable versus non-cleavable
Cleavable linkers are prized for their versatility while non-cleavable for their enhanced safety. However, the first kind of linker chemistry remains dominant for therapeutic ADC applications. Their versatility ensures the possibility of controlled payload release while ensuring the ADC remains relatively stable in circulation.
A downside of their versatility is the increased risk of off-target toxicity that can be mitigated by engineering conjugation sites and reducing their exposure to solvents and, thus, maximizing their stability in plasma. Many cleavable ADCs have been shown to forgo the need for internalization to exert a therapeutic effect. These linkers often cleave in the tumor’s microenvironment (higher acidity and oxidative stress), making them more effective when dealing with large solid tumors often impermeable to large antibodies.
When targeting hematological tumors, non-cleavable ADCs could be more useful considering their high specificity and low toxicity. For this reason, the use of one chemistry or the other remains dependent on the type and progression of cancer in each patient. Experts have argued that the concerted action of the two chemistries could have a synergistic effect by hastening the elimination of different types of cancer cells in the same patient.
The advantages and limitations of each chemistry can be summarized in the table below.
| Cleavable linkers | Non-cleavable linkers | |
|---|---|---|
| Advantages |
|
|
| Limitations |
|
|
Concluding remarks
Choosing between cleavable and non-cleavable linker chemistry has a dramatic impact on ADC tolerance and therapeutic efficiency. Cleavable linkers remain the dominant type of chemistry for most applications due to their enhanced versatility – a wider range of compatibility with different drugs and antigens. However, they have a higher risk of exerting off-target toxicity which can be mitigated by employing engineering strategies that protect linkers from premature exposure and cleavage.
Due to their versatility, cleavable linkers comprise a widely diversified class of well-established linkers with proven clinical efficiency. In contrast, the development of novel non-cleavable linkers has lagged due to the inherent difficulties in matching linkers with payloads without significantly modifying the structure and activity of the latter.
Despite their limitations, many experts believe both kinds of linker chemistry can be used for synergistic ADC-based treatments. Since at least 99% of cancer cells need to be eliminated for cancer to go into remission, it is important to harness the advantages of both types of linkers to achieve an optimal therapeutic effect.
- Baah, S. et al. Antibody–Drug Conjugates—A Tutorial Review. Molecules. 2021; 26:2943. doi: 10.3390/molecules26102943
- Bargh, J. D. et al. Cleavable linkers in antibody-drug conjugates. Chem Soc Rev. 2019; 48(16):4361-4374. doi: 10.1039/c8cs00676h
- Lu, J. et al. Linkers Having a Crucial Role in Antibody–Drug Conjugates. Int J Mol Sci. 2016; 17(4): 561. doi: 10.3390/ijms17040561
- Tsuchikama, K. and An, Z. Antibody-drug conjugates: recent advances in conjugation and linker chemistries. Protein Cell. 2018; 9(1): 33–46. doi: 10.1007/s13238-016-0323-0
You could also be interested in: