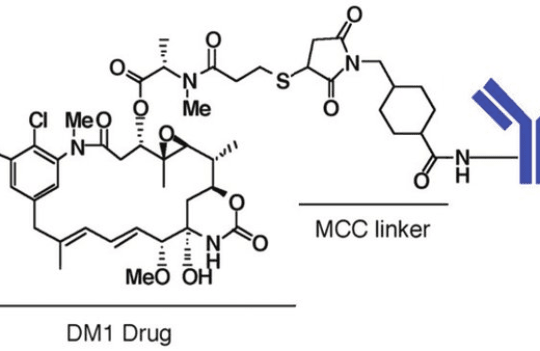
Antibody-drug conjugates
Antibody-drug conjugates
How to select the right antibody carrier for ADCs?
Monoclonal antibodies are essential precursors of ADCs. Choosing the proper antibody for the conjugate often dictates its therapeutic efficiency. Several properties must be taken into account during the early stage of antibody selection for ADCs including target affinity and specificity, internalization rate, and stability or aggregation, among others. In this article, we will go through the most important factors that must be considered to guarantee optimal efficiency and help you select the best antibody carrier for your ADC applications.
Typical monoclonal antibodies used for ADC generation belong to the IgG class. These large glycoproteins (150 kDa) are used in conjugation with small cytotoxic drugs (e.g., calicheamicin at 1368 Da) that cannot be used as stand-alone chemotherapeutics. ADCs are one of the fastest-growing classes of biopharmaceuticals for oncology, for this reason, much of the work and research on these compounds has focused on the identification of highly specific cancer markers and antibody engineering for enhanced surface-marker affinity.
However, as more and more ADCs enter the clinical pipeline, it is important to revise the knowledge acquired on antibody selection and optimization to expand the use of these compounds for other applications.
Vital properties of an antibody precursor for ADC generation
In comparison to conventional monoclonal therapies, ADCs face a higher attrition rate in the clinical pipeline due to their less substantial track record and higher toxicity. For this reason, many experts believe that to improve the current success rate of ADCs and expand their applicability, it is urgent to test multiple targets and antibody-payload conjugations to select the most effective ones. In other words, it is urgent to optimize the monoclonal antibody alongside its conjugated form for optimal therapeutic efficiency.
Three critical properties need to be screened and optimized to consider an antibody suitable for ADC development. The first is the antibody’s affinity and specificity towards antigen-positive tumor cells; the second its internalization rate; and third, its subsequent intracellular localization.
These properties are important because, to produce a therapeutic effect, conventional ADCs need to target specific tumor cells, be internalized via lysosomes, and undergo degradation to release their cytotoxic cargo. In general, the tighter the antibody binds to the antigen and the faster its internalization, the more efficient the ADC therapy.
However, the relationship between these factors and therapeutic efficiency is not always linear, and antibodies with slow internalization rates have been found to work for some applications (e.g., Kadcyla™). Moreover, solid tumors have been shown to be particularly recalcitrant to ADC due to the slow diffusion rates and antibody trapping by peripheral cancer cells (antigen barrier). Recent studies suggest non-internalizing ADCs may be more suitable for the treatment of these tumors because they release the cargo in the extracellular space and that cargo can more easily diffuse through the tumor. In this case, the antibody carrier should be resistant to internalization and the linker compatible with fast cleavage in the proximity of cancer cells.
How to optimize an antibody carrier for ADCs?
As a general rule, affinity, internalization, and localization play a crucial role in ADC efficiency. For this reason, screening/profiling antibodies (natural or engineered) based on these properties during early discovery is a robust approach to antibody optimization.
These properties can be measured using the following methods:
Antibody affinity
Affinity can be directly measured or estimated using multiple techniques including ELISA or flow cytometry. However, since most ADC target surface markers, it is vital to measure affinity using an antigen’s native conformation (surface-bound).
For this reason, techniques that allow measuring the affinity to cells versus purified proteins should be preferred. One technique able to achieve this is Surface Plasmon Resonance imaging (SPRi). Using label-free optical detection, SPRi can measure the interaction between an antibody with its ligand expressed in native or recombinant cells.
Antibody internalization
Internalization can be measured using different techniques including flow cytometry often in combination with surface quenching and studies with radiolabeled antibodies. Other suitable methodologies are based on imaging such as macro-confocal imaging. Alternatively, the direct and indirect measurement of an ADC cytotoxicity (in vitro assays) is also a reliable way to estimate its internalization by tumor cells. However, the latter is less useful for early antibody discovery as it requires the prior conjugation of the carrier with its toxic cargo.
To bypass the need for ADC synthesis and allow carrier discovery and optimization, assays relying on flow cytometry or confocal microscopy are typically preferred.
Antibody location
Conventional internalizing ADCs need to undergo degradation before the cytotoxic cargo can be released to the intracellular environment. For this reason, it is extremely useful to monitor the location of an antibody or corresponding conjugate once it is internalized by the cell. Colocalization experiments (confocal microscopy or flow cytometry) are the most well-established methodologies to determine the fate of a carrier or ADC upon internalization.
Critical factors for target selection of ADC antibodies
Due to the high toxicity of ADC cargoes, proper target selection is one of the most important steps in the development of these biopharmaceuticals. Several factors should be considered when selecting a target including specificity, expression levels, internalization, heterogeneity, and accessibility. Much of the information required to choose a suitable target for an ADC comes from basic and functional research on disease markers.
Specificity is at the core of every successful ADC biopharmaceutical. For cancer treatments, antibodies should bind strictly to antigens enriched on the surface of tumors (high expression levels). At the same time, these antigens should either have low expression in other tissues or be present only in expendable tissues or those with the ability to regenerate quickly.
Another important property is heterogeneity. Intra-patient target heterogeneity is particularly widespread in cancer. For instance, only 20% of breast cancer patients are HER2-positive (human epidermal growth factor receptor 2), which would limit the efficiency of a HER2-targeting ADC to a subset of the population and require the use of additional differential diagnostic assays to determine which patients would better benefit from this type of treatment. Unfortunately, it isn’t always possible to select a universal target for a specific disease. For this reason, experts believe it will be increasingly important to divide patients into subgroups (i.e., personalized medicine) and develop specific therapies for each.
Both the choice of an antibody carrier and target antigen influence ADC internalization. Ideally, for conventional internalizing ADCs, the antigen must be able to internalize ADCs and quickly be recycled to replenish its levels on the surface of the tumor cell. The antigen should also be easily accessible to the ADC. For instance, solid tumors are harder to target than hematogenous tumors (i.e., “liquid” tumors) as the former hinders the penetration of antibodies, ADCs, and even small drugs. The bulkier the tumor, the harder it is to treat it with ADC therapy, independently of the expression level of a specific antigen.
Concluding remarks
The optimization of an antibody carrier for ADC development should take several factors into account. Although these antibodies by themselves do not need to elicit a therapeutic response, they must be able to bind tightly to a tumor or cell-specific surface marker and, ideally, undergo quick internalization and degradation via lysosomes or to quickly release their cargoes on the periphery of solid tumors.
For this reason, antibody profiling during early ADC development is vital to improve the success rate of these biopharmaceuticals. Thus, SPRi, confocal microscopy, and flow cytometry remain useful techniques to screen antibodies or antibody libraries to determine the best carrier for each specific ADC application.
- Bander, N. H. Antibody-drug conjugate target selection: critical factors. Methods Mol Biol. 2013; 1045:29-40. doi: 10.1007/978-1-62703-541-5_2
- Harper, J. et al. Selecting an optimal antibody for antibody-drug conjugate therapy: internalization and intracellular localization. Methods Mol Biol. 2013; 1045:41-49. doi: 10.1007/978-1-62703-541-5_3
You could also be interested in: