 CAR-T cell therapy
CAR-T cell therapy
CAR-T cell engineering: strategies to overcome remaining limitations
How can CAR-T Cell engineering overcome the remaining limitations of cell therapy?
CAR-T cell engineering has become one of the most active areas of cancer immunotherapy research. By genetically modifying T cells to express synthetic chimeric antigen receptors, researchers can redirect immune cells toward tumor-associated antigens and trigger targeted cytotoxic activity.
CAR-T therapies have already transformed the treatment of several hematological malignancies, especially relapsed or refractory B-cell cancers and multiple myeloma. However, major challenges remain, including antigen escape, limited efficacy in solid tumors, manufacturing complexity, and treatment-related toxicities such as cytokine release syndrome and neurotoxicity.
Today, the field is moving beyond first-generation design principles. New strategies now focus on multi-antigen targeting, improved trafficking, enhanced persistence, armored CAR-T cells, gene-edited allogeneic platforms, in vivo CAR-T generation, and more precise safety control mechanisms.
This article summarizes how CAR-T cell engineering is helping overcome the current limitations of cell therapy and expanding its therapeutic potential.
- CAR-T cell engineering: Where to start?
- Recognition
- Trafficking
- Proliferation and persistency
- Tumor microenvironment
- Control mechanisms
- CAR-T cell engineering: a closer look at current limitations and opportunities
- Reduce antigen escape
- Improving the treatment of solid tumors
- Reducing systemic toxicity
- Concluding remarks
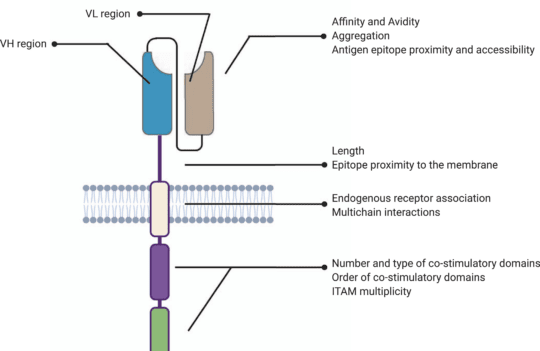
Chimeric antigen receptors (CARs) modulate natural T cell receptors (TCR) and are typically transduced/transfected into T cells sourced from autologous or allogeneic origins. This strategy has proven to be remarkably efficient at treating certain types of B cell cancers (via the CD19 antigen), namely B cell leukemia or lymphoma. CARs are characterized by an ectodomain (antigen-binding and spacer regions), a transmembrane domain, and an endodomain vital for triggering the signaling cascade upon encountering a tumor-associated antigen and subsequent cytotoxic response.
Despite the encouraging clinical success, CAR-T cells still face many challenges tied to excessive toxicity, limited anti-tumor activity, antigen escape, restricted trafficking, and reduced efficacy against solid tumors. Due to these challenges, CAR-T cell engineering is currently an active field of research where new solutions are regularly developed to improve safety, efficacy (including against solid tumors), cost, manufacturability, and availability.
CAR-T cell engineering: Where to start?
There is no single solution to improve CAR-T cell therapies. Instead, successful CAR-T cell engineering requires optimization across several interconnected parameters, from antigen recognition to cell persistence, tumor infiltration, resistance to the tumor microenvironment, and safety control.
Current engineering efforts can be grouped into five major areas:
Recognition
Antigen recognition remains one of the most decisive factors in CAR-T cell efficacy. The extracellular binding domain, often an scFv, must recognize the target antigen with the right balance of specificity, affinity, and functional activity.
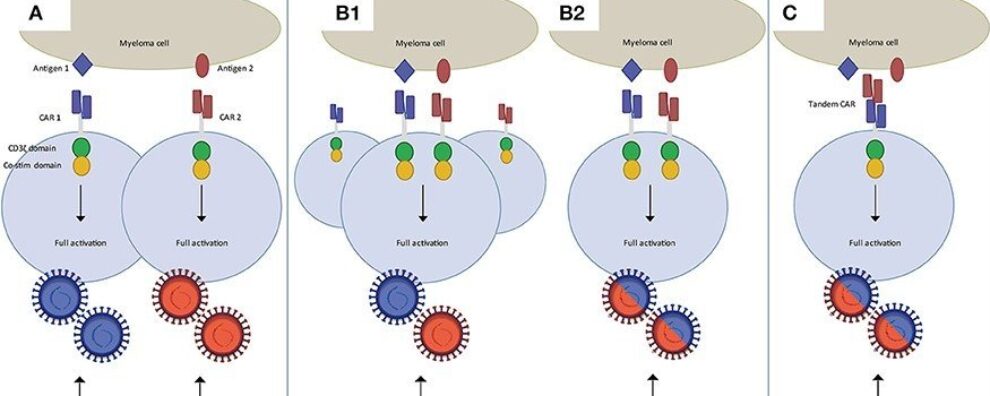
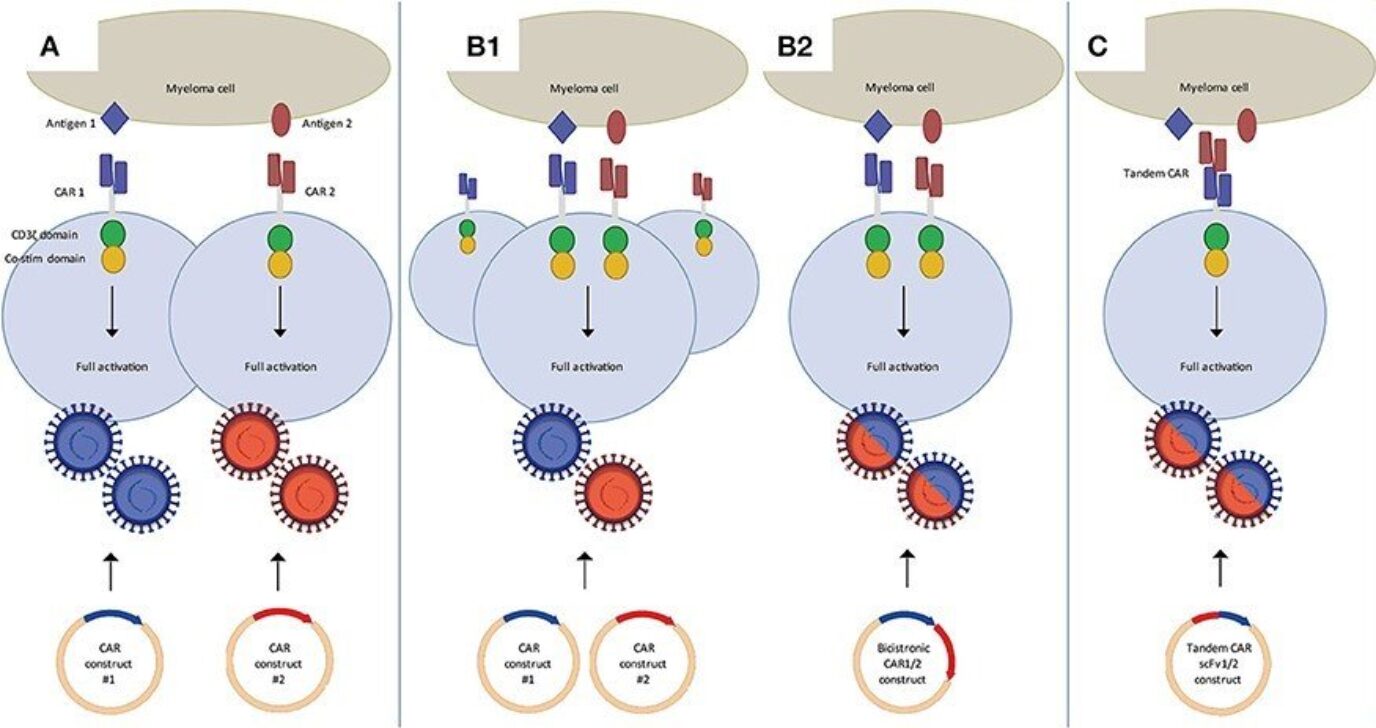
Early CAR-T therapies demonstrated strong clinical results in CD19-positive hematological malignancies, but antigen loss or downregulation can lead to relapse. To reduce this risk, researchers are developing dual-targeting CAR-T cells, tandem CARs, bicistronic CARs, and logic-gated CAR systems capable of recognizing more than one antigen.
The quality of the binding domain is also critical. Fully human or humanized binders can help reduce immunogenicity, while affinity tuning can improve selectivity and reduce off-tumor toxicity. For challenging tumor-associated targets, screening binders directly on cells can help identify candidates that recognize the native antigen conformation.
Discover our services:

Trafficking
CAR-T cell trafficking refers to the ability of engineered T cells to reach the tumor site. This is usually less problematic in hematological malignancies, where cancer cells circulate or reside in accessible compartments such as blood, bone marrow, or lymphoid tissues.
In solid tumors, trafficking remains a major barrier. CAR-T cells must migrate toward the tumor, cross physical barriers, penetrate dense extracellular matrices, and remain active in a hostile microenvironment.
To improve trafficking, current strategies include engineering CAR-T cells to express chemokine receptors matching the chemokine profile of the tumor, using locoregional delivery, and combining CAR-T cells with approaches that remodel the tumor stroma. These strategies are particularly important for extending CAR-T therapy beyond blood cancers.

Proliferation and persistency
CAR-T cell proliferation and persistence strongly influence therapeutic durability. A CAR-T product must expand after infusion, maintain cytotoxic activity, and survive long enough to eliminate tumor cells.
Modern CAR design now pays close attention to intracellular signaling domains. CD28-based CARs are often associated with rapid activation, while 4-1BB-based CARs are generally linked to longer persistence and memory-like phenotypes. Newer designs are also exploring cytokine signaling modules, transcriptional rewiring, and gene-edited integration sites to improve consistency and reduce exhaustion.
Manufacturing also plays a major role. T-cell fitness, culture duration, expansion methods, and product composition can all affect clinical performance. This is why next-generation CAR-T development increasingly combines receptor design with optimized production workflows.
Discover our services:
Antibody engineering services
AI-driven antibody developability improvement service
Tumor microenvironment
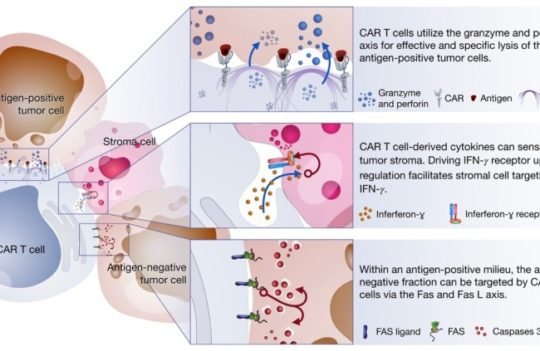
Many solid tumors secret a plethora of biomolecules that modulate the microenvironment, often leading to the downregulation or inhibition of T cells. For this reason, engineering CAR-T cells able to resist this suppressive pressure may be instrumental for the use of these cells against solid tumors.

Control mechanisms
CAR-T cells are known to cause, on occasion, excessive off-tumor and off-target toxicity. For this reason, engineering strategies that allow more precise control over the process in vivo could be instrumental to increase the safety of these cells. One of such strategies already in the test phase is the introduction of molecular switches. These signals allow clinicians to shut down or switch on the cytotoxic response by administering chemical signals like small drugs and thus making these therapies safer.

CAR-T cell engineering: a closer look at current limitations and opportunities
Reduce antigen escape
Antigen escape occurs when tumor cells lose or downregulate the target antigen recognized by CAR-T cells. This is one of the main mechanisms of relapse after single-antigen CAR-T treatment.
Multi-target CAR-T strategies are being developed to reduce this risk. These include:
- co-administration of two CAR-T cell populations
- dual CAR-T cells expressing two independent CARs
- tandem CAR-T cells containing two binding domains in one receptor
- logic-gated CAR-T cells requiring specific antigen combinations
The success of these approaches depends heavily on target selection. Ideal antigen combinations should provide broad tumor coverage, stable expression, and limited overlap with healthy tissues.
This is where early-stage antibody discovery and antigen validation become critical. Identifying binders that recognize tumor-relevant conformations, including membrane proteins or peptide-HLA complexes, can help create more specific and durable CAR-T candidates.
Interestingly, many tumor-associated antigens are also expressed at low levels on normal cells and tissue. For this reason, dual-targeting might not suffice to ensure CAR-T cell specificity. For this reason, researchers are tuning antigen-binding domains to recognize specific tumor-associated post-translational modifications (PTM). This strategy is useful because solid tumors are known to overexpress truncated O-glycans. In this way, scFvs that target these modified glycans are already being investigated at the preclinical stage. They are expected to further reduce the risk of antigen escape while minimizing the off-target toxicity of CAR-T cell treatments.
Improving the treatment of solid tumors
Solid tumors remain one of the most important frontiers for CAR-T therapy. While hematological cancers have shown strong responses, solid tumors present additional layers of complexity: heterogeneous antigen expression, poor immune cell infiltration, physical stromal barriers, and suppressive tumor microenvironments.
Current engineering strategies aim to address these barriers through:
- improved antigen selection
- multi-antigen targeting
- chemokine receptor engineering
- armored CAR-T cells
- local or regional delivery
- resistance to suppressive pathways such as TGF-β or PD-1/PD-L1 signaling
- combination with checkpoint blockade or tumor microenvironment-modulating agents
Recent clinical and preclinical advances suggest that CAR-T therapy for solid tumors is progressing, but the field still requires careful optimization of target biology, CAR design, delivery route, and safety mechanisms.
Reducing systemic toxicity
CAR-T cell therapies can cause powerful immune activation. While this is part of their antitumor mechanism, excessive activation can lead to systemic toxicity.
The most clinically relevant toxicities include cytokine release syndrome, immune effector cell-associated neurotoxicity syndrome, on-target/off-tumor toxicity, prolonged cytopenias, and inflammatory complications.
Engineering strategies to reduce toxicity include:
- optimizing scFv affinity to avoid activation by low-antigen-density healthy tissues
- selecting costimulatory domains according to expected antigen density and desired activation profile
- using human or humanized binding domains to reduce immunogenicity
- incorporating suicide switches or drug-regulated control systems
- using logic-gated CARs to improve tumor selectivity
- exploring local administration to reduce systemic exposure
The next generation of CAR-T therapies will likely rely on a combination of better target selection, safer receptor architecture, improved manufacturing, and built-in control mechanisms.
Concluding remarks
CAR-T cell therapy has already changed the treatment landscape for several hematological malignancies. However, the future of the field depends on overcoming persistent limitations such as antigen escape, poor solid tumor efficacy, complex manufacturing, and systemic toxicity.
Modern CAR-T cell engineering is now moving toward more sophisticated and controllable systems. Multi-target CARs, armored CAR-T cells, allogeneic platforms, in vivo engineering, and safety switches are expanding what these therapies can achieve.
For researchers and biotech teams, successful CAR-T development begins with the right antigen, the right binder, and the right engineering strategy. From antigen design and scFv discovery to CAR construct development, integrated workflows can help accelerate the identification of stronger CAR-T candidates.
- Zugasti I, Espinosa-Aroca L, Fidyt K, et al. CAR-T cell therapy for cancer: current challenges and future directions. Signal Transduction and Targeted Therapy. 2025;10:210.
https://doi.org/10.1038/s41392-025-02269-w - Li J, Liu C, Zhang P, et al. Optimizing CAR T cell therapy for solid tumours: a clinical perspective. Nature Reviews Clinical Oncology. 2025;22:953–968.
https://doi.org/10.1038/s41571-025-01075-1 - Escobar G, Berger TR, Maus MV. CAR-T cells in solid tumors: Challenges and breakthroughs. Cell Reports Medicine. 2025;6(11):102353.
https://doi.org/10.1016/j.xcrm.2025.102353 - Diorio C, Teachey DT, Grupp SA. Allogeneic chimeric antigen receptor cell therapies for cancer: progress made and remaining roadblocks. Nature Reviews Clinical Oncology. 2025;22:10–27.
https://doi.org/10.1038/s41571-024-00959-y - Bot A, Scharenberg A, Friedman K, et al. In vivo chimeric antigen receptor (CAR)-T cell therapy. Nature Reviews Drug Discovery. 2026;25:116–137.
https://doi.org/10.1038/s41573-025-01291-5